The US Senate on Tuesday passed a sweeping industrial policy bill aimed at countering the surging economic threat from China, overcoming partisan divisions to support pumping more than US$170 billion into research and development.
The measure cleared the chamber on a 68-32 vote, one of the most significant bipartisan achievements in the US Congress since US President Joe Biden took office in January.
It also represents the largest investment in scientific research and technological innovation “in generations,” Senate Majority Leader Chuck Schumer said.

Photo: Reuters
The bill now heads to the US House of Representatives, which earlier passed a different version.
The two will have to be reconciled into a single bill before it is sent to the White House for the president’s signature.
Biden said he was “encouraged” by the Senate’s passage of the US Innovation and Competition Act.
“We are in a competition to win the 21st century, and the starting gun has gone off,” Biden said. “As other countries continue to invest in their own research and development, we cannot risk falling behind. America must maintain its position as the most innovative and productive nation on Earth.”
The package, a key provision of which addresses a shortage of semiconductors that has slowed production at US automakers this year, would help US industry bolster its capacity and improve technology.
It is seen as crucial for US efforts to avoid being outmaneuvered by Beijing as the adversaries compete in the race to technological innovation.
“Today, the Senate took a critical bipartisan step forward to make the investments we need to continue America’s legacy as a global leader in innovation,” US Secretary of Commerce Gina Raimondo said in a statement. “This funding isn’t just about addressing the current semiconductor chip shortage, it is about long-term investments.”
Schumer called the measure “one of the most important things this chamber has done in a very long time, a statement of faith in America’s ability to seize the opportunities of the 21st century.”
The proposal aims to address a number of technological areas in which the US has fallen behind China.
The bill allocates US$52 billion for a previously approved plan to increase domestic manufacturing of semiconductors.
It also authorizes US$120 billion over five years for activities at the US National Science Foundation to advance priorities, including research and development in key areas such as artificial intelligence and quantum science.
It facilitates tie-ups between private firms and research universities.
“This is an opportunity for the United States to strike a blow on behalf of answering the unfair competition that we are seeing from communist China,” said US Senator Roger Wicker, one of the bill’s main cosponsors.
“For everything from national security to economic policy, there’s a clear and urgent need to reorient the way our country views and responds to the challenge from China,” US Senator John Cornyn said.
In Beijing, officials yesterday accused Washington of “paranoid delusion” after the bill advanced.
The foreign affairs committee of China’s National People’s Congress said that the bill was “full of Cold War mentality and ideological prejudice.”
“The bill shows that the paranoid delusion of egoism has distorted the original intention of innovation and competition,” Xinhua news agency reported it as saying.
It is an attempt to interfere in China’s internal affairs, and deprive it of its “legitimate right to development through technology and economic decoupling,” it reported.

A signaling system malfunction disrupted high-speed rail (HSR) services beginning at 8am today, with trains temporarily reduced to three northbound and three southbound trains per hour as authorities conduct inspections. The malfunction occurred on a section of track in Miaoli County during pre-operation checks early this morning, forcing northbound and southbound trains to use a single track, the HSR operator said. The regular schedule has been replaced with three hourly trains offering only nonreserved seating in each direction, stopping at every station, it said, adding that business class cars would still have reserved seating. Departures from terminal stations are scheduled at the top

DRONE CENTRAL: Taiwan aims to become Asia’s democratic hub for drones, with most exports focused on high-quality military-grade models, an official said Taiwan’s drone industry is expected to expand significantly by 2030, producing 100,000 units per month and exporting half of them, the Ministry of Economic Affairs said yesterday. Current drone production capacity is about 15,000 units per month, but the industry can quickly scale up as demand increases, Industrial Development Administration Director-General Chiou Chyou-huey (邱求慧) told a news conference in Taipei. Taiwan’s drone output grew 2.5-fold last year to NT$12.9 billion (US$408.3 million) under a government program to develop the uncrewed vehicle sector, he said. The Executive Yuan in October last year approved plans to invest NT$44.2 billion into domestic production of uncrewed aerial

VERBOSE VESSELS: A CGA cutter and a China Coast Guard exchanged verbal barbs for more than a day in Taiwanese-controlled waters before the Chinese vessel left The Taiwanese and Chinese coast guards had a standoff near the strategically located Pratas Islands (Dongsha Islands, 東沙群島) in the north of the South China Sea, the Coast Guard Administration (CGA) said yesterday. The two sides engaged in intense radio exchanges over sovereignty claims during the 33-hour standoff. China Coast Guard vessel 3501 eventually left the restricted waters, 26.6 nautical miles (49.2km) west of the Pratas Islands, at 5pm yesterday, the CGA said. Lying approximately between southern Taiwan and Hong Kong, the Taiwan-controlled Pratas are seen by some security experts as vulnerable to Chinese attack due to their distance — more than
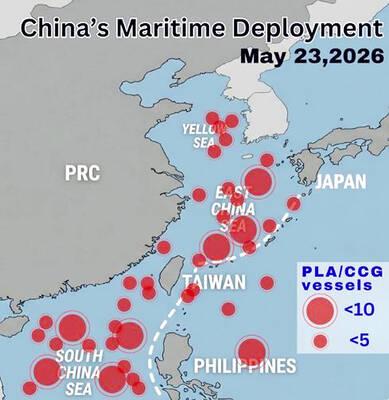
WARNING: China should stop engaging in actions that undermine regional peace and stability, as it would only build resentment among people across the Strait, the CGA said China has deployed more than 100 navy, coast guard and other vessels in waters from the Yellow Sea to the South China Sea and the western Pacific since US President Donald Trump and Chinese President Xi Jinping (習近平) met in Beijing, National Security Council Secretary-General Joseph Wu (吳釗燮) said yesterday. “In this part of the world, #China is the one & only PROBLEM wrecking the #StatusQuo & threatening regional peace & stability,” Wu wrote on X. In a separate post, he said Beijing was coercing Taiwan’s maritime domain, calling it illegal and provocative, after the Coast Guard Administration (CGA) expelled a