The process through which Taichung County-based biotech company Adimmune Corp came to manufacture its A(H1N1) influenza vaccine was flawed and paved the way for safety concerns that have surrounded the vaccine, a US expert on infectious diseases told the Taipei Times this week.
Ho Mei-shang (何美鄉), a research fellow at Academia Sinica’s Institute of Biomedical Sciences and former director of biotechnology research at Adimmune, disputes this view, however, saying in an interview on Tuesday that the vaccine is safe and that there was nothing wrong with the process.
The Centers for Disease Control (CDC) announced on July 13 that Adimmune, the sole bidder, had won a government contract to produce 5 million doses of the (A)H1N1 vaccine, at NT$199 per dose, for delivery by the end of October. By early this month, Adimmune had provided 6.95 million doses, while Switzerland-based Novartis had provided 2.02 million (at about NT$400 per dose), out of the government’s purchases of 10 million and 5 million doses respectively.
The government has spent NT$3 billion (US$94.4 million) purchasing vaccines from Adimmune.
The problems begin when looking at the chain of events that led to the awarding of the contract, the US expert said, speaking on condition of anonymity because of the sensitive nature of her position.
As early as May 5, the CDC said that Adimmune, in cooperation with the National Health Research Institutes, would manufacture a domestic influenza vaccine. This was more than a month before a WHO survey of global flu vaccine production capacity on July 7 concluded that countries such as Taiwan that had not placed early orders for the vaccine would not get any vaccine after November.
Adimmune’s chairman is Steve Chan (詹啟賢), deputy secretary-general of the Chinese Nationalist Party (KMT). Chan was also the deputy executive director of President Ma Ying-jeou’s (馬英九) presidential campaign.
For decades a producer of animal vaccines, Adimmune is the only firm in Taiwan with the capacity to produce human vaccines.
Prior to the A(H1N1) contract, it had never produced human vaccines on its own. All it did, the source said, was “package” vaccines made by other, mostly Japanese, companies.
Ho maintained, however, that through close cooperation with Japanese and Dutch vaccine makers, Adimmune “leapfrogged” and obtained the know-how to make human vaccines.
Most vaccine manufacturers, including GSK and Novartis, have decades of experience producing seasonal flu vaccines. While H1N1 is only a variant of the seasonal flu, major manufacturers usually obtain licensure from the US Food and Drug Administration (FDA) to expedite the process based on regulations regarding “strain change.”
Regardless, manufacturers providing H1N1 vaccines to the US have undergone clinical trials, most of which are sponsored by the National Institutes of Health (NIH).
EXPEDITED PROCESS
Adimmune, which had no experience producing the vaccine, should never have been allowed to enter an “expedited process,” the source said.
Adimmune only conducted a small-scale clinical trial with less than 500 subjects, far less than the 5,000 to 6,000 that are usually required for new vaccines.
Ho said that as the vaccine is based on the seasonal flu vaccine, it is not a new vaccine and therefore there was “no point” in conducting more rigorous testing.
The source, however, said that as Adimmune had never produced the seasonal flu vaccine, the Department of Health (DOH) should have demanded the company conduct more clinical trials.
“This is the scientific foundation to claim that a vaccine is ‘safe,’” the source said. “You can’t cut corners on this. Science requires replicability.”
In response, Ho said that, “lack of experience does not jeopardize quality. It just means more costs.”
Adimmune’s chairman has said that clinical trials are a “redundant procedure for bureaucrats,” adding that most Western countries had shortened them to speed up vaccinations.
In other words, despite its lack of experience, the company felt confident it could produce a safe product with few trials, while more experienced manufacturers that also entered an “expedited process” proceeded more cautiously — GSK did 6,340 tests, Novartis did 4,768 and the NIH conducted 3,951, data shows.
(The Novartis version of the vaccine sold to Taiwan is a H5N1 markup with an adjuvant, which was purchased via a special procurement as the adjuvant has yet to be approved in Taiwan.)
WHO guidelines for vaccine manufacturing show that three demo batches of a vaccine, as well as pre-approval inspection, must be completed before a vaccine can be released.
The source said Adimmune finished the clinical study on Oct. 21 and obtained the licensure on Nov. 12. The nationwide vaccine program started on Nov. 15.
“According to the timeline, Adimmune couldn’t have made it in three weeks,” the source said. “This means they mass produced before an approval [license] was granted,” the source said.
Ho confirmed this was the case.
“Adimmune was taking a chance,” Ho said, because if it failed to obtain approval, it would have “to discard all batches that had been produced beforehand.”
“I was among the very few top management team and I was adamant about going ahead to produce the vaccine as at that time, there appeared to [be] no other opportunity to secure H1N1 vaccines for ... Taiwan,” Ho said.
“My intention at the time was that we, the Taiwanese people ... would lose more if there was no H1N1 vaccine at all. Thus, with a certain degree of confidence in what Adimmune could achieve, the risk of failure was deemed slim and we went ahead,” Ho said.
“There was no ethical issue. It was all about regulation and it was OK as long as approval was obtained eventually,” she said.
“Because the approval was based on the inspection and documentation of consistency of three consecutive [vaccine] batches — even though the paperwork was completed later — it was retroactively applicable,” she said.
On Jan. 7, the DOH said autopsies showed that six of 17 deaths possibly related to the vaccine were due to other causes, with investigations continuing on the other 11.
Ho confirmed that all cases involved individuals who had received the Adimmune vaccine, adding that this didn’t mean the vaccine was less safe than Novartis’, as the majority of vaccines administered here were Adimmune’s.
The source also saw problems with the DOH.
ETHICS
“Again and again, DOH officials have sided with Adimmune, something I have never seen in the US,” the source said. “If the commissioner of the [US] FDA, the head of the [US] CDC or the NIH endorsed a certain brand, they not only would be in clear violation of ethics codes, they would be committing political suicide.”
When a legislator asked to monitor the egg production process (influenza vaccines are produced in fertilized chicken eggs), Adimmune said “it’s a commercial secret,” the source said. “Later, when the CDC head [Steve Kuo, 郭旭崧] was asked how the sterility of the eggs was assured, his answer was: ‘Adimmune examined it.’”
“Was there no DOH oversight?” the source asked.
Ho maintains that from the very beginning, the DOH kept a professional distance from Adimmune. There was no dialogue between Adimmune and the CDC, which was responsible for buying the vaccine, she said.
Despite Adimmune’s requests, the DOH also hasn’t allowed it to release clinical test results.
The reason, Ho said, is that “technically speaking, the clinical trial has not been finished.”
The contract stipulates that until then, no information can be disclosed.
“The DOH adamantly opposes disclosure,” Ho said. “That makes me think something is fishy.”
For the source, the vaccine mess is another example of the gambling attitude of the Ma administration, conflict of interest and lack of transparency.
“As a federal government employee with experience in issuing contracts, and as a medical professional, I would say that if the same process occurred in the US, not only would it [make] the headlines in the Washington Post, but criminal investigations would follow,” she said.
For Ho, the problem was poor government communication and media speculation. Chan’s role as chairman also made it easier to politicize the matter, she said.
“Adimmune is more than a private company — it does public health,” she said. “The firm could have gone bankrupt, but everybody worked hard to make it happen in time and provide Taiwan with a vaccine production facility.”
Asked if she’d taken the vaccine, Ho said, “Yes, — Adimmune’s.”

FUKUOKA SITUATION: Japanese media reported that the pathogen is expected to be identified by the summer, while the CDC downplayed the idea that it was hMPV A “mysterious cold-like illness” reported in Japan’s Fukuoka Prefecture does not seem to be a new disease, but Japanese authorities have been asked about the situation, the Centers for Disease Control (CDC) said yesterday. The Fukuoka Prefectural Medical Association on Wednesday told a news conference that a “mystery cold” that has become a hot topic on social media is “highly likely to be caused by some kind of viral infection,” Japan’s KBC News reported. “Many people are experiencing symptoms starting with a sore throat, followed by a runny nose, phlegm and a severe cough,” KBC News reported, citing association officials. Health authorities are

Nvidia Corp CEO Jensen Huang (黃仁勳) arrived in Taiwan yesterday ahead of upcoming AI and technology events, saying he plans to meet with clients and Taiwan Semiconductor Manufacturing Co Chairman C.C. Wei (魏哲家) during his visit. After landing at Taipei Songshan Airport, Huang posed for photos with fans and handed out Yakult drinks to reporters and supporters waiting at the scene, saying he has “a lot to do” during the trip. Asked about reports that Nvidia’s planned headquarters site in Taipei’s Beitou Shilin Technology Park could break ground on May 27, Huang said that if the company holds an event, he would
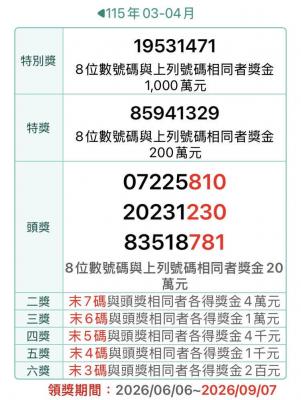
The Ministry of Finance this afternoon announced the winning numbers for the March-April uniform invoice lottery. The winning number for the NT$10 million (US$318,060) special prize is 19531471, and the winning number for the NT$2 million grand prize is 85941329. Three numbers were drawn for the NT$200,000 first prize: 07225810, 20231230 and 83518781. Those with receipts matching the last seven digits of any of the first-prize numbers will win the NT$40,000 second prize, while those matching the last six digits will win the NT$10,000 third prize. Those whose receipts match the last five digits of the first-prize numbers can claim the NT$4,000 fourth prize,

Taiwan Travelogue (臺灣漫遊錄), which earlier this week became the first Taiwanese novel to win the International Booker Prize, is to be adapted into a television series through a Taiwan-Japan coproduction, producer Chang Chen-yu (張辰漁) said yesterday. Chang, a producer at World Softest Production Film Co, wrote on Facebook that the company had been searching for projects with international appeal that retain a strong Taiwanese identity after colleagues and Japanese partners strongly recommended the novel. After reading the book, Chang said he immediately decided to pursue the screen rights. “A great story has the power to transcend time and borders, and connect countless people,”