The Center for Disease Control (CDC) has ordered 190,000 doses of avian influenza vaccine to enhance the nation's flu-fighting capability amid renewed pandemic warnings, a senior official said yesterday.
According to CDC Deputy Director Chou Chih-hao (周志浩), the government decided to procure the vaccine from two foreign suppliers after a new resistant strain of the highly pathogenic H5N1 virus deadly to humans and poultry was found to have spread through Asia.
Chou quoted a report by scientists in Hong Kong and the US as saying that the new virus -- dubbed the "Fujian-like" strain -- may have mutated in response to vaccination programs designed to halt the disease in farm flocks.
Noting that the strain emerged last year and has already spread throughout China as well as to Hong Kong, Thailand, Laos and Malaysia, the report said that the spread of this virus over such a large geographical region within a short period of time directly challenges current disease control measures.
Against this backdrop, Chou said the CDC has spent nearly NT$60 million (US$1.81 million) purchasing 190,000 doses of the vaccine against the H5N1 virus from two foreign companies.
The vaccine is 70 percent to 80 percent effective and has a shelf life of between six months and two years, Chou said.
Nevertheless, Chou added that although the vaccine has been proven safe and effective in preventing avian influenza in humans in initial clinical tests, it has not yet been approved for commercial sale.
Therefore, Chou said, the vaccine will be stockpiled for emergency epidemic control use or domestic clinical testing.
"Basically, only frontline medical and epidemic control personnel will get vaccinated," Chou said, adding that the vaccine will not be available to members of the public.
According to the procurement contracts, Chou said, the vaccine will be delivered by the end of this year.
"The vaccine will be ready for use if an avian flu outbreak occurs this winter," he said.
If each medical professional gets two doses of the vaccine, Chou said, the stockpile will be enough to cover 90,000 individuals, or one-third of the country's health care personnel.
Moreover, Chou said, the CDC has also kept in stock enough doses of Tamiflu -- an oral anti-viral drug for treatment of influenza -- for 2.3 million people, or 10 percent of the nation's population in accordance with the recommendation of the WHO.
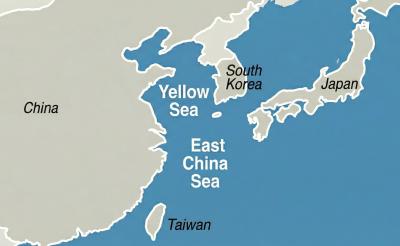
China has reserved offshore airspace in the Yellow Sea and East China Sea from March 27 to May 6, issuing alerts usually used to warn of military exercises, although no such exercises have been announced, the Wall Street Journal (WSJ) reported yesterday. Reserving such a large area for 40 days without explanation is an “unusual step,” as military exercises normally only last a few days, the paper said. These alerts, known as Notice to Air Missions (Notams), “are intended to inform pilots and aviation authorities of temporary airspace hazards or restrictions,” the article said. The airspace reserved in the alert is

More than 6,000 Taiwanese students have participated in exchange programs in China over the past two years, despite the Mainland Affairs Council’s (MAC) “orange light” travel advisory, government records showed. The MAC’s publicly available registry showed that Taiwanese college and university students who went on exchange programs across the Strait numbered 3,592 and 2,966 people respectively. The National Immigration Agency data revealed that 2,296 and 2,551 Chinese students visited Taiwan for study in the same two years. A review of the Web sites of publicly-run universities and colleges showed that Taiwanese higher education institutions continued to recruit students for Chinese educational programs without

A bipartisan group of US senators has introduced a bill to enhance cooperation with Taiwan on drone development and to reduce reliance on supply chains linked to China. The proposed Blue Skies for Taiwan Act of 2026 was introduced by Republican US senators Ted Cruz and John Curtis, and Democratic US senators Jeff Merkley and Andy Kim. The legislation seeks to ease constraints on Taiwan-US cooperation in uncrewed aerial systems (UAS), including dependence on China-sourced components, limited access to capital and regulatory barriers under US export controls, a news release issued by Cruz on Wednesday said. The bill would establish a "Blue UAS

The Republic of China Army Command yesterday relieved Kinmen Defense Battalion commander after authorities indicted the officer on charges connected to using methamphetamine. The Kinmen District Prosecutors’ Office on Wednesday detained Colonel He (何) after the Coast Guard linked him to drug shipments and proceeded to charge him yesterday for using and possessing crystal meth. The man was released on a NT$50,000 bail and banned from leaving Kinmen, the office said. Army Chief of Staff Lieutenant General Chen Chien-yi (陳建義) told a news conference yesterday that He has been removed and another officer is taking over the unit as the acting commander. The military