The Animal and Plant Health Inspection Agency on Saturday said that draft amendments to regulations on human medicines for veterinary use would allow pre-stocking of medicines by veterinarians to ensure animals’ medical rights.
The Regulations Governing the Use and Management of Human Drugs on Dogs, Cats and Non-economic Animals (人用藥品使用於犬貓及非經濟動物之使用管理辦法) announced in 2024 would officially take effect in July.
The agency has announced 701 pharmaceuticals for human use that can be applied to dogs, cats or non-farm animals, but only 216 are registered as animal medicines eligible for veterinary use.

Photo courtesy of a reader
Many pet owners and veterinarians are worried that more than 500 medicines yet to be registered might not be legitimately stocked at veterinary clinics, given that only registered medicines can be supplied by pharmaceutical companies directly to veterinarians.
An online platform set up by Sincere Animal Hospital superintendent and veterinarian Liu Jung-tsung (劉榮宗) has listed four issues that would impact the medical rights of pet owners as well as pets and other non-farm animals.
First, some lifesaving medicines such as diuretics, which are often used to treat heart disease, are yet to be registered by any pharmaceutical company.
Second, medical oxygen, which is indispensable to anesthetic operations and critically ill patients requiring supplemental oxygen, is also yet to be registered.
Third, pharmaceutical manufacturers are facing increased costs due to registration fees, label revision and extra warehousing, which could push up pharmaceutical prices.
Fourth, pet owners must get a prescription from a veterinarian to exchange for unregistered medicines from pharmacies, meaning they cannot obtain large quantities for preparation purposes.
In response to concerns, agency deputy director-general Fu Hsueh-li (傅學理) said a meeting with relevant agencies would be held on Friday with amendments to the regulations being planned to permit pre-stocking of such medicines by veterinarians and establish a convenient supply mechanism for medical gases.
Industrial oxygen should also be legally banned from being used to substitute for medical oxygen in veterinary facilities, he said.
Meanwhile, Federation of Taiwan Pharmacists deputy secretary-general Lee Yi-hsuan (李懿軒) said that although the registration rate remained low, pharmaceutical companies should not be blamed, as the agency did not hold a policy briefing with the industry until Feb. 9.
Taiwan Veterinary Medical Association chairman David Tan (譚大倫) yesterday added that many pharmaceutical operators are yet to register their pharmaceutical products as animal medicines, mainly because changing label information and packaging would increase costs.
Hiring veterinary pharmacists would also largely increase both the costs for veterinary clinics and their charges to pet owners, given the small scale of most domestic animal hospitals, he said.
Critically, the current training system for pharmacists is solely focused on human healthcare and does not provide separate training for veterinary medications, he said.

The Centers for Disease Control (CDC) yesterday reported the first case of a new COVID-19 subvariant — BA.3.2 — in a 10-year-old Singaporean girl who had a fever upon arrival in Taiwan and tested positive for the disease. The girl left Taiwan on March 20 and the case did not have a direct impact on the local community, it said. The WHO added the BA.3.2 strain to its list of Variants Under Monitoring in December last year, but this was the first imported case of the COVID-19 variant in Taiwan, CDC Deputy Director-General Lin Ming-cheng (林明誠) said. The girl arrived in Taiwan on

South Korea is planning to revise its controversial electronic arrival card, a step Taiwanese officials said prompted them to hold off on planned retaliatory measures, a South Korean media report said yesterday. A Yonhap News Agency report said that the South Korean Ministry of Foreign Affairs is planning to remove the “previous departure place” and “next destination” fields from its e-arrival card system. The plan, reached after interagency consultations, is under review and aims to simplify entry procedures and align the electronic form with the paper version, a South Korean ministry official said. The fields — which appeared only on the electronic form
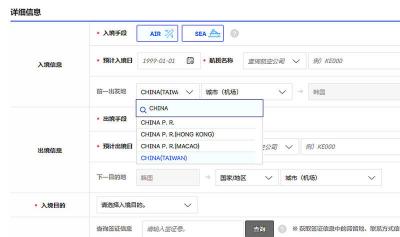
The Ministry of Foreign Affairs (MOFA) is suspending retaliation measures against South Korea that were set to take effect tomorrow, after Seoul said it is updating its e-arrival system, MOFA said today. The measures were to be a new round of retaliation after Taiwan on March 1 changed South Korea's designation on government-issued alien resident certificates held by South Korean nationals to "South Korea” from the "Republic of Korea," the country’s official name. The move came after months of protests to Seoul over its listing of Taiwan as "China (Taiwan)" in dropdown menus on its new online immigration entry system. MOFA last week

A bipartisan group of US senators has introduced a bill to enhance cooperation with Taiwan on drone development and to reduce reliance on supply chains linked to China. The proposed Blue Skies for Taiwan Act of 2026 was introduced by Republican US senators Ted Cruz and John Curtis, and Democratic US senators Jeff Merkley and Andy Kim. The legislation seeks to ease constraints on Taiwan-US cooperation in uncrewed aerial systems (UAS), including dependence on China-sourced components, limited access to capital and regulatory barriers under US export controls, a news release issued by Cruz on Wednesday said. The bill would establish a "Blue UAS