Phase 2 clinical trial results of a COVID-19 vaccine developed by Medigen Vaccine Biologics Corp generated good data, experts said, although they warned that there was still no way to know how effectively it would prevent the disease.
“Data look quite good,” Academia Sinica researcher Michael Lai (賴明詔) said on Thursday after Medigen announced that its vaccine candidate had shown no major safety concerns.
The data showed that the vaccine produced sufficient neutralizing antibodies and as long as there were antibodies, there should be protection, Lai said, adding that its toxicity was low.

Photo: Tsai Wen-chu, Taipei Times
The Medigen vaccine has achieved the goals of creating sufficient neutralizing antibodies and having low toxicity, but only phase 3 trials could determine if it actually protects people who receive it, said Lai, who is a virologist and a coronavirus researcher.
There are many ways to produce vaccines, including via protein subunits such as the one Medigen is working on, he said.
The most traditional way is to use the virus to produce a vaccine with several proteins that the immune system recognizes should not be there, triggering an immune response, he said.
However, this method can produce a level of toxicity, he said.
A protein subunit vaccine has only one protein, in this case a spike protein, he said.
“It is purer, but could trigger a weaker immune reaction,” Lai said. “Different vaccine production methods have different advantages and disadvantages. One cannot be compared to another.”
It could take several years for a domestic vaccine to go through phase 3 trials, so given Taiwan’s situation, “it should opt for second-best,” even though major vaccines made elsewhere in a matter of months obtained enough efficacy data in phase 3 trials to get emergency use authorization (EUA), he said.
It would be acceptable for Medigen to apply for EUA with only the results of its phase 2 trials, he said.
National Taiwan University Hospital pediatrician Lu Chun-yi (呂俊毅), who is secretary-general of the Taiwan Immunization Vision and Strategy organization, said that the Medigen data were “good.”
Almost every participant had antibodies after being inoculated and the antibody concentration data were promising, Lu said.
However, there is no international consensus on how high the geometric mean titer (GMT) — or antibody concentration — has to be to indicate sufficient protection, he said.
Moreover, no uniform, globally accepted method for verifying antibody concentrations exists, meaning that vaccines have a wide range of GMTs, from as low as just over 100 to above 3,000, he said.
Different laboratories conduct trials with different materials and methods, leading to results that vary across labs, Lu said.
Shih Shin-ru (施信如), who heads the Research Center for Emerging Viral Infections at Chang Gung University said that it was still too early to say whether Medigen had successfully developed a COVID-19 vaccine, because nobody knows how high the neutralizing antibody concentration needs to be.
Medigen said it would seek an EUA from the Food and Drug Administration as soon as possible based on the promising results from its phase 2 clinical trials.
It would also apply to the European Medicines Agency and other international health authorities to begin phase 3 clinical trials as soon as possible, it said.

The Centers for Disease Control (CDC) yesterday reported the first case of a new COVID-19 subvariant — BA.3.2 — in a 10-year-old Singaporean girl who had a fever upon arrival in Taiwan and tested positive for the disease. The girl left Taiwan on March 20 and the case did not have a direct impact on the local community, it said. The WHO added the BA.3.2 strain to its list of Variants Under Monitoring in December last year, but this was the first imported case of the COVID-19 variant in Taiwan, CDC Deputy Director-General Lin Ming-cheng (林明誠) said. The girl arrived in Taiwan on

South Korea is planning to revise its controversial electronic arrival card, a step Taiwanese officials said prompted them to hold off on planned retaliatory measures, a South Korean media report said yesterday. A Yonhap News Agency report said that the South Korean Ministry of Foreign Affairs is planning to remove the “previous departure place” and “next destination” fields from its e-arrival card system. The plan, reached after interagency consultations, is under review and aims to simplify entry procedures and align the electronic form with the paper version, a South Korean ministry official said. The fields — which appeared only on the electronic form

A bipartisan group of US senators has introduced a bill to enhance cooperation with Taiwan on drone development and to reduce reliance on supply chains linked to China. The proposed Blue Skies for Taiwan Act of 2026 was introduced by Republican US senators Ted Cruz and John Curtis, and Democratic US senators Jeff Merkley and Andy Kim. The legislation seeks to ease constraints on Taiwan-US cooperation in uncrewed aerial systems (UAS), including dependence on China-sourced components, limited access to capital and regulatory barriers under US export controls, a news release issued by Cruz on Wednesday said. The bill would establish a "Blue UAS
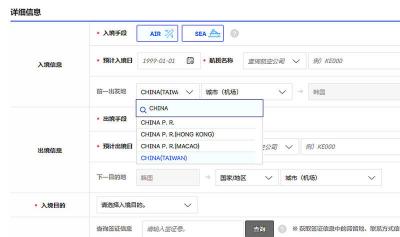
The Ministry of Foreign Affairs (MOFA) is suspending retaliation measures against South Korea that were set to take effect tomorrow, after Seoul said it is updating its e-arrival system, MOFA said today. The measures were to be a new round of retaliation after Taiwan on March 1 changed South Korea's designation on government-issued alien resident certificates held by South Korean nationals to "South Korea” from the "Republic of Korea," the country’s official name. The move came after months of protests to Seoul over its listing of Taiwan as "China (Taiwan)" in dropdown menus on its new online immigration entry system. MOFA last week