The Chinese Nationalist Party (KMT) yesterday filed a lawsuit against Minister of Health and Welfare Chen Shih-chung (陳時中) and Food and Drug Administration (FDA) Director Wu Shou-mei (吳秀梅) alleging corruption in the decision to issue an emergency use authorization (EUA) for the COVID-19 vaccine developed by the Medigen Vaccine Biologic Corp (高端疫苗生物製劑).
The FDA had bypassed standard procedures and, in broad defiance of public and international opinion, decided to approve the vaccine, the KMT said.
Citing FDA data, KMT caucus secretary-general Cheng Li-wun (鄭麗文) said that Medigen started producing vaccines last month, before it received the EUA, which is evidence that the company was given assurances that its vaccine would be approved and made available for use by next month.

Photo: CNA
“We have become the only nation to approve an EUA for a vaccine that has not passed phase 3 clinical trials,” Cheng said.
Medigen yesterday said that it had received approval to conduct phase 3 clinical trials on 1,000 people in Paraguay.
KMT Chairman Johnny Chiang (江啟臣) cited the vaccine approval process in the US, saying that the process is recorded and that its review processes are transparent.
Taiwan, on the other hand, had not recorded the review process, he said, adding that the public does not know who attended the meeting, or what kind of opposition was voiced.
“We have set the lowest bar for vaccine approval worldwide,” Chiang said.
KMT Disciplinary Committee director Yeh Ching-yuan (葉慶元), who is the party’s legal representative, said that the procedures to approve domestic vaccines went against past practice, despite Taiwan having access to international vaccines and having no immediate need for them.
The FDA was guilty of corruption as it forced through the approval of the EUA by replacing half of the academics on the panel of specialists, Yeh said.
In response, Democratic Progressive Party caucus secretary-general Lo Chih-cheng (羅致政) said that the legislature would declassify and make public the minutes of the specialists’ meeting within two weeks.
At the specialists’ meeting, 18 members voted to approve the vaccine, one voted for further discussion after asking for more information and one voted to not approve the vaccine, Wu said on Monday, adding that the convener of the meeting was not allowed to vote.
Under the EUA, the company must provide a safety report every month, she said.

A signaling system malfunction disrupted high-speed rail (HSR) services beginning at 8am today, with trains temporarily reduced to three northbound and three southbound trains per hour as authorities conduct inspections. The malfunction occurred on a section of track in Miaoli County during pre-operation checks early this morning, forcing northbound and southbound trains to use a single track, the HSR operator said. The regular schedule has been replaced with three hourly trains offering only nonreserved seating in each direction, stopping at every station, it said, adding that business class cars would still have reserved seating. Departures from terminal stations are scheduled at the top

Taiwan is still in the process of assessing the possibility of recruiting workers from Eswatini, the Ministry of Foreign Affairs said yesterday, adding that its goal is to help Eswatini upgrade its vocational training centers. If there are plans to recruit workers from Eswatini, safeguarding national security, protecting public health and ensuring the employment rights of Taiwanese would be prerequisites, Department of West Asian and African Affairs Director-General Yen Chia-liang (顏嘉良) told a news conference. Key considerations would also include filling labor shortages in specific industries, and fostering bilateral professional and technical exchanges, he said. Yen was asked about the progress of labor

VERBOSE VESSELS: A CGA cutter and a China Coast Guard exchanged verbal barbs for more than a day in Taiwanese-controlled waters before the Chinese vessel left The Taiwanese and Chinese coast guards had a standoff near the strategically located Pratas Islands (Dongsha Islands, 東沙群島) in the north of the South China Sea, the Coast Guard Administration (CGA) said yesterday. The two sides engaged in intense radio exchanges over sovereignty claims during the 33-hour standoff. China Coast Guard vessel 3501 eventually left the restricted waters, 26.6 nautical miles (49.2km) west of the Pratas Islands, at 5pm yesterday, the CGA said. Lying approximately between southern Taiwan and Hong Kong, the Taiwan-controlled Pratas are seen by some security experts as vulnerable to Chinese attack due to their distance — more than
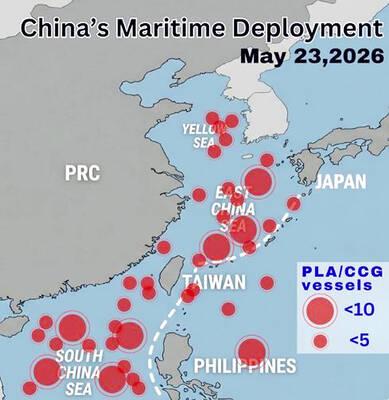
WARNING: China should stop engaging in actions that undermine regional peace and stability, as it would only build resentment among people across the Strait, the CGA said China has deployed more than 100 navy, coast guard and other vessels in waters from the Yellow Sea to the South China Sea and the western Pacific since US President Donald Trump and Chinese President Xi Jinping (習近平) met in Beijing, National Security Council Secretary-General Joseph Wu (吳釗燮) said yesterday. “In this part of the world, #China is the one & only PROBLEM wrecking the #StatusQuo & threatening regional peace & stability,” Wu wrote on X. In a separate post, he said Beijing was coercing Taiwan’s maritime domain, calling it illegal and provocative, after the Coast Guard Administration (CGA) expelled a