A two-drug combination pill dramatically reduced deaths among blacks with heart failure, a landmark finding that is expected to lead to government approval of the first medication marketed for a specific race.
Black cardiologists hailed this form of racial profiling after years in which minorities got short shrift in medical studies. Others complained that the drug also might help whites and should have been tested in them, but wasn't for business reasons.
"At times you can't win," said Dr. Augustus Grant, past president of the Association of Black Cardiologists, which supported the study. "Here we have a wonderful trial that shows a clear result and the issue is raised, `Why was this trial only done in African Americans?'"
The nationwide study is the largest ever done solely on blacks with heart failure. The findings were reported Monday at an American Heart Association meeting in New Orleans and will be published tomorrow in the New England Journal of Medicine. The drug's maker plans to seek Food and Drug Administration approval by the end of the year.
Heart failure affects 5 million Americans, but blacks are 2 1/2 times more likely to develop it. It happens when the heart is too weak to pump effectively, causing fluid to back up in the lungs and leaving people weak and short of breath. Half die within five years of diagnosis.
Earlier research suggested that standard heart failure drugs called ACE inhibitors do not work as well in blacks, and that blacks may have lower amounts of nitric oxide, which plays many roles in heart health, in their blood.
Two chemicals -- isosorbide dinitrate and hydralazine -- boost this substance, but administering the right dose is complicated when they are prescribed separately. A Massachusetts biotechnology company, NitroMed, developed a combination pill, BiDil, that gets around this problem, but the FDA refused to license it as a new drug because earlier studies involving mostly white patients who got the chemicals separately showed no benefit.
But there were tantalizing signs that BiDil helped the few blacks in the studies, and NitroMed won a patent to use it just in that minority group.
The company then launched a study of 1,050 blacks. Half of them got standard heart failure drugs and a placebo; the other half got those drugs plus BiDil. The study was stopped ahead of schedule last year when doctors saw BiDil clearly was better.
After roughly two years' use, only 6.2 percent of the patients who took BiDil had died versus 10.2 percent who got only standard heart failure drugs. That translates to a 43 percent reduction in deaths, said Dr. Anne Taylor of the University of Minnesota, one of the study's leaders.
Only 16.4 percent on BiDil required hospitalization for heart failure, versus 24.4 percent of the rest.
BiDil had substantial side effects -- 47.5 percent on it had headaches, compared with 19.2 of the others. Dizziness occurred in 29.3 percent on the drug and 12.3 percent on fake pills.
But the favorable overall result "virtually ensures FDA approval," because the agency previously told the company that a successful study in blacks would merit it, Dr. M. Gregg Bloche, a Georgetown University lawyer and Johns Hopkins University physician, said in a commentary in the medical journal.
That is "cause for celebration" for blacks, but the company now will have no financial incentive to do a larger trial in whites because its "black-only" patent allows it to sell it that way and keep generic versions from coming the market until 2020, he said.
Others worried that the drug might not be the best choice for every black but that they will automatically be prescribed "the black pill" solely on the basis of skin color.
Being black is not a black-and-white distinction, said Dr. Timothy Gardner of the University of Pennsylvania in Philadelphia, who had no role in the study. "Physiologically, it's a sort of continuous variable," including people of mixed races, he said.

‘IN A DIFFERENT PLACE’: The envoy first visited Shanghai, where he attended a Chinese basketball playoff match, and is to meet top officials in Beijing tomorrow US Secretary of State Antony Blinken yesterday arrived in China on his second visit in a year as the US ramps up pressure on its rival over its support for Russia while also seeking to manage tensions with Beijing. The US diplomat tomorrow is to meet China’s top brass in Beijing, where he is also expected to plead for restraint as Taiwan inaugurates president-elect William Lai (賴清德), and to raise US concerns on Chinese trade practices. However, Blinken is also seeking to stabilize ties, with tensions between the world’s two largest economies easing since his previous visit in June last year. At the
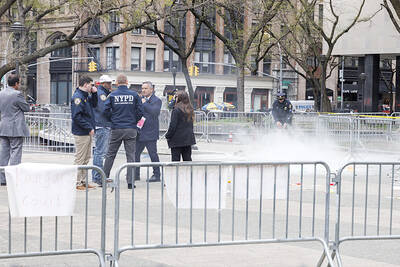
UNSETTLING IMAGES: The scene took place in front of TV crews covering the Trump trial, with a CNN anchor calling it an ‘emotional and unbelievably disturbing moment’ A man who doused himself in an accelerant and set himself on fire outside the courthouse where former US president Donald Trump is on trial has died, police said yesterday. The New York City Police Department (NYPD) said the man was declared dead by staff at an area hospital. The man was in Collect Pond Park at about 1:30pm on Friday when he took out pamphlets espousing conspiracy theories, tossed them around, then doused himself in an accelerant and set himself on fire, officials and witnesses said. A large number of police officers were nearby when it happened. Some officers and bystanders rushed

Beijing is continuing to commit genocide and crimes against humanity against Uyghurs and other Muslim minorities in its western Xinjiang province, U.S. Secretary of State Antony Blinken said in a report published on Monday, ahead of his planned visit to China this week. The State Department’s annual human rights report, which documents abuses recorded all over the world during the previous calendar year, repeated language from previous years on the treatment of Muslims in Xinjiang, but the publication raises the issue ahead of delicate talks, including on the war in Ukraine and global trade, between the top U.S. diplomat and Chinese

RIVER TRAGEDY: Local fishers and residents helped rescue people after the vessel capsized, while motorbike taxis evacuated some of the injured At least 58 people going to a funeral died after their overloaded river boat capsized in the Central African Republic’s (CAR) capital, Bangui, the head of civil protection said on Saturday. “We were able to extract 58 lifeless bodies,” Thomas Djimasse told Radio Guira. “We don’t know the total number of people who are underwater. According to witnesses and videos on social media, the wooden boat was carrying more than 300 people — some standing and others perched on wooden structures — when it sank on the Mpoko River on Friday. The vessel was heading to the funeral of a village chief in