Concerned groups yesterday called for a more reasonable prices for both patented and generic drugs.
"On average, pharmaceutical companies spend US$800 million developing an innovative drug before putting it on the market and they have only eight years to make a profit out of it [before generic versions are allowed]," said Hsieh Yen-yau (
Hsieh said the Research, Development and Evaluation Commission and the Ministry of Audit should cooperate on setting price guidelines for drugs.
He also suggested the government take into consideration the prices of medicines abroad.
Hsieh said that both generic and patented drugs were necessary given the difficulty of raising national health insurance fees.
He emphasized, however, that the Bureau of Food and Drug Analysis must ensure that the ingredients in drugs have come from reliable sources.
He said that generic drugs must follow national standards to avoid problems.
Meanwhile, he proposed that patients be given the right to choose their own drugs as long as they are willing to pay the price difference between a generic and a more expensive brand-name drug.
Hsieh made the statement at a press conference that was hosted by the Association of International Research-Based Pharmaceutical Manufacturers.
The association also released results of a study by Yeh Ching-yin (
The study involved a survey of 20 Taiwanese doctors who are leaders in medical associations, and reported that 85 percent said name-brand drugs were safer and more reliable.
Seventy percent said that they see treatment quality decline when they switch from prescribing name-brand drugs to generic ones.

The military has spotted two Chinese warships operating in waters near Penghu County in the Taiwan Strait and sent its own naval and air forces to monitor the vessels, the Ministry of National Defense (MND) said. Beijing sends warships and warplanes into the waters and skies around Taiwan on an almost daily basis, drawing condemnation from Taipei. While the ministry offers daily updates on the locations of Chinese military aircraft, it only rarely gives details of where Chinese warships are operating, generally only when it detects aircraft carriers, as happened last week. A Chinese destroyer and a frigate entered waters to the southwest
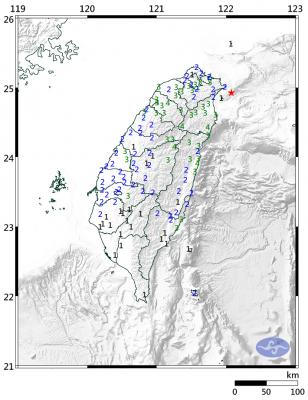
A magnitude 6.1 earthquake struck off the coast of Yilan County at 8:39pm tonight, the Central Weather Administration (CWA) said, with no immediate reports of damage or injuries. The epicenter was 38.7km east-northeast of Yilan County Hall at a focal depth of 98.3km, the CWA’s Seismological Center said. The quake’s maximum intensity, which gauges the actual physical effect of a seismic event, was a level 4 on Taiwan’s 7-tier intensity scale, the center said. That intensity level was recorded in Yilan County’s Nanao Township (南澳), Hsinchu County’s Guansi Township (關西), Nantou County’s Hehuanshan (合歡山) and Hualien County’s Yanliao (鹽寮). An intensity of 3 was

Japanese Prime Minister Sanae Takaichi’s comment last year on Tokyo’s potential reaction to a Taiwan-China conflict has forced Beijing to rewrite its invasion plans, a retired Japanese general said. Takaichi told the Diet on Nov. 7 last year that a Chinese naval blockade or military attack on Taiwan could constitute a “survival-threatening situation” for Japan, potentially allowing Tokyo to exercise its right to collective self-defense. Former Japan Ground Self-Defense Force general Kiyofumi Ogawa said in a recent speech that the remark has been interpreted as meaning Japan could intervene in the early stages of a Taiwan Strait conflict, undermining China’s previous assumptions

Taiwan Railways Corp (TRC) today announced that Shin Kong Mitsukoshi has been selected as the preferred bidder to operate the Taipei Railway Station shopping mall, replacing the current operator, Breeze Development Co Ltd. Among eight qualified firms that delivered presentations and were evaluated by a review committee, Shin Kong Mitsukoshi was ranked first, while Breeze was named the runner-up, the rail company said in a statement. Contract negotiations are to proceed in accordance with regulations, it said, adding that if negotiations with the top bidder fail, it could invite the second-ranked applicant to enter talks. Breeze in a statement today expressed doubts over