The Department of Health appealed yesterday to the public to temporarily refrain from using drugs containing phenylpropanolamine (PPA) after the US took steps to remove PPA from all drug products on Monday.
"We need to understand more about the side effects of PPA and let the public know whether Taiwan will ban PPA or not," said Oliver Hu (
The US Food and Drug Administration issued a public health advisory concerning the risk of hemorrhagic stroke -- or bleeding into the brain -- associated with PPA hydrochloride. Scientists at Yale University School of Medicine conducted a study in which researchers found an association between PPA use and strokes in women. An increased risk of hemorrhagic stroke in the three days after taking the medicine was detected among women using the drug for weight control and for nasal decongestion. Men may also be at risk.
Hu said European countries and Japan are also considering the matter, but have not yet followed America in banning drugs with PPA.
"In the interests of public health, we appeal to the public not to use drugs containing PPA before the department's research comes out," he stressed.
According to the department's statistics, there are around 460 drugs for weight loss and colds containing PPA. In the past, all approved diet drugs contained PPA.
The department held a press conference yesterday to announce the approval of a new medicine, Orlistat, as a prescription diet drug that contains no PPA.
"This drug is not over-the-counter and has its side effects. But many people are interested in it. That's why we'd like to remind people to use it with care," said Lee Ming-liang (
Lee said Orlistat can be used for up to two years, but patients need to replenish Vitamins A, D, E and K while taking the drug.
Pregnant women, the elderly and people whose liver, kidney or digestive functions are weak, or whose bile secretion is insufficient are advised not to use Orlistat, Lee said.
"Over 90 percent of users will have gastrointestinal problems and their excrement will become very oily," said Larry Ho (
Ho said the resultant flatulence and anal seepage when using the drug could also lead potentially to hygiene problems. The function of Orlistat is to prevent the intestines and stomach from absorbing fat from food. Unabsorbed oil is then excreted from the body.
Ho said clinical experiments in other countries had been conducted to prove the effect of Orlistat. Forty-one people in Taiwan have also tested it. "Average weight loss was around 4kg," Ho said. "But diet control, as well as the drug, plays an important role in losing weight."
"Its optimum effect is to reduce 10 percent of body weight," Ho said. "Any medicine which claims to lose more than 10 percent of weight is misleading customers."

‘NO SECURITY RISK’: The Railway Bureau reassured the public that the technicians’ activities were limited to technical guidance and did not involve sensitive systems The Railway Bureau yesterday said it had invited eight Chinese technicians to assist with an airport MRT construction project. The bureau issued the confirmation after an Internet user said Chinese nationals had entered the construction zone of Taiwan Taoyuan International Airport’s Terminal 3 project. They asked why “individuals from an enemy state” were allowed access to such a major national infrastructure project, which raised serious concerns over Taiwan’s industrial safety, sensitive systems and information security. The bureau’s Northern Region Engineering Branch Office said subcontractor Taiwan Handle Industrial Co (台灣手把工業) of the Taoyuan airport MRT’s “Contract No. CU05 Project A14 Station Civil, MEP &

A US uncrewed surface vessel (USV) encountered multiple Chinese warships during an autonomous transit of the Taiwan Strait, US defense company Seasats said in a statement on Wednesday. Seasats announced that a Lightfish USV had completed the first autonomous transit of the Taiwan Strait. Over five days, the USV traversed the entire length of the Strait while constantly monitoring surface vessel traffic, the company said. The Lightfish encountered multiple Chinese warships, one of which was a Chinese People’s Liberation Army Navy (PLAN) Type 056 corvette, it said. The Chinese vessels were operating “well within Taiwan’s exclusive economic zone without transmitting their identity via the

‘BOOMING’: ’ The number of partners we have here is incredible. You can see from their stock prices. They’re doing so well, they’re so happy,’ Jensen Huang said Nvidia Corp’s spending in Taiwan has ballooned to about US$150 billion a year, 10 times the US$10 billion to US$15 billion the company spent five years ago, Nvidia chief executive officer Jensen Huang (黃仁勳) said yesterday, suggesting Taiwan’s strategic importance in the global artificial intelligence (AI) supply chain. “Taiwan is the epicenter of the AI revolution. This is where the chips come, packaging comes. This is where the systems are made. This is where AI supercomputers were created,” Huang said at a meeting for the company’s employees in Beitou-Shilin Technology Park (北投士林科技園區) in Taipei, the planned site of Nvidia’s Taipei headquarters. “Taiwan
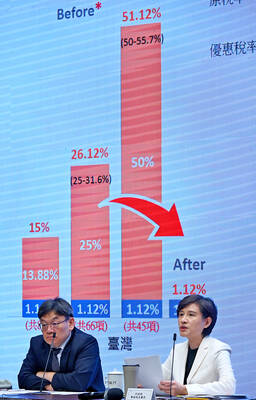
GREATER REACH? Auto parts and wood products would face tariffs of up to 15%, matching those targeting the EU, Japan and South Korea, Vice Premier said The US has announced that preferential tariff treatment for Taiwan’s non-semiconductor Section 232 goods would take effect retroactively from May 1, the Executive Yuan said yesterday. The US government yesterday posted a notice on the Federal Register’s public inspection Web site previewing tariff concessions for Taiwan under a memorandum of understanding (MOU) on Taiwan-US investment after two months of negotiations. The MOU signed on Jan. 15 stipulated three major preferential tariff arrangements: a 15 percent “reciprocal” tariff rate for Taiwan without stacking most-favored nation (MFN) rates; preferential Section 232 treatment for semiconductors and related products; and preferential Section 232 treatment for non-semiconductor