New drug review processes would be accelerated and completed in 120 days in response to a US plan to raise pharmaceutical tariffs, the Ministry of Health and Welfare said yesterday.
Food and Drug Administration (FDA) Deputy Director-General Wang Der-yuan (王德原) yesterday told a news conference that measures have been taken to optimize new medicine review processes and help pharmaceutical companies obtain drug permit licenses as soon as possible.
Such processes normally take about 360 days to complete, if the new medicine is a new chemical entity (NCE) — a drug with new chemicals as principal components — or a drug made from biological sources, also known as biologics.

Photo: AP
However, NCEs or biologics considered important for pediatric treatments and serious diseases, as well as breakthrough therapies can have their review prioritized, with the process shortened to 240 days, he said.
If an NCE or a biologic has been approved in two of the US, the EU or Japan, it is qualified for a “type 1 express review” and can have its review completed as fast as in 180 days, Wang said.
If it has been approved in the three areas with identical data on chemistry, manufacturing and controls, it is qualified for a “type 2 express review” and its review process could be completed in 120 days, he added.
The ministry would also continue to encourage pharmaceutical companies to diversify sources of bulk drugs or active pharmaceutical ingredients so that they would not rely on a single supplier, he said.
China and India are the two major sources of bulk drugs used in Taiwan’s pharmaceutical industry, he said.
The FDA has requested pharmaceutical companies to evaluate whether they should find more bulk drug sources, Wang said, adding that the agency would prioritize the review of new medicines relying on a single source.
Pharmaceutical companies should also use domestically made bulk drugs, although they are mostly more expensive and less adopted, he said.
The administration has compiled an inventory of all US-made pharmaceutical products and sent a notice to their permit license holders to fully understand the medicine supply chain situation and properly boost domestic medicine stocks, he said.
It would also check stocks of primary medicines, such as anti-cancer drugs, orphan drugs and biologics to promptly initiate an investigation and evaluation whenever there is a potential shortage, he said.
The FDA would ask manufacturers of substitute medicines to increase production, or launch a drug import or manufacturing project, if necessary, he said.
Pharmaceutical companies can propose price adjustments to stabilize the market if changes in the global supply chain cause a surge in drug production costs, he said.
The ministry would continue to promote the application of generic drugs and biosimilars — biologics with similar structure and functions of a reference biologic made by another company — to bolster domestically made new medicines, Wang added.
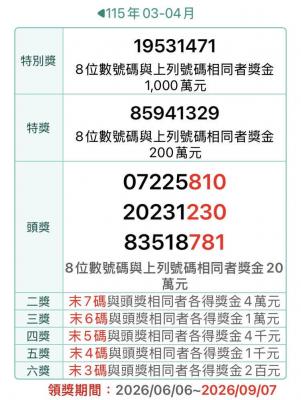
The Ministry of Finance this afternoon announced the winning numbers for the March-April uniform invoice lottery. The winning number for the NT$10 million (US$318,060) special prize is 19531471, and the winning number for the NT$2 million grand prize is 85941329. Three numbers were drawn for the NT$200,000 first prize: 07225810, 20231230 and 83518781. Those with receipts matching the last seven digits of any of the first-prize numbers will win the NT$40,000 second prize, while those matching the last six digits will win the NT$10,000 third prize. Those whose receipts match the last five digits of the first-prize numbers can claim the NT$4,000 fourth prize,

SIX SUBSIDIES: The monthly allowance for older farmers is to increase to NT$10,000, and NT$5,000 is to be given to homemakers under the national pension system, Lai said The government is to implement major welfare policies for disadvantaged groups, including raising the monthly allowance for older farmers to NT$10,000 and providing homemakers with NT$5,000 per month, President William Lai (賴清德) said yesterday. Lai made the remarks during a visit to Wangling Temple in Chiayi County, saying that the planned increases were being introduced amid economic growth and an increase in tax revenue. Touting a policy, in which the government plans to provide a monthly allowance of NT$5,000 for every child under the age of 18 in a bid to address Taiwan’s low birthrate, Lai said that if received for the
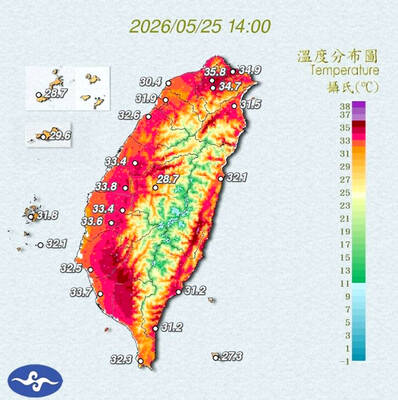
STAY COOL: The HPA recommended that people stay hydrated, use air-conditioning or fans while indoors, wear loose-fitting clothes and walk in the shade while outdoors Employers must implement measures such as installing cooling equipment, and providing drinking water and rest breaks for outdoor workers starting from Monday next week, the Taipei Department of Labor said on Sunday. Employers who fail to comply could face fines of NT$30,000 to NT$300,000 under the Occupational Safety and Health Act (職業安全衛生法), the department said. Businesses in Taipei employing fewer than 100 workers, as well as registered self-employed workers with labor insurance coverage, could receive on-site assessments and guidance from occupational safety consultants to help them apply for central government subsidies to implement or improve heat-protection measures, it said. Under the Ministry of
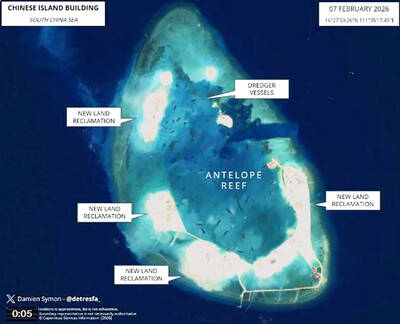
ISOLATION: The outposts would serve as support and backup bases, forcing US forces to either face China head-on or reroute, increasing travel time and operational costs China’s outposts in the South China Sea could be used to delay and constrain foreign forces during a conflict in the Taiwan Strait, giving Beijing a critical window to carry out amphibious landing and blockade operations, a report said. The Chinese Communist Party’s (CCP) forward operating bases on islands and reclaimed features in the South China Sea could delay foreign forces long enough for the Chinese People’s Liberation Army (PLA) to secure a key 48-to-72-hour window in the Taiwan Strait, a report commissioned by the Mainland Affairs Council found. The report, conducted by the Institute for National Defense and Security Research, examined