The Centers for Disease Control (CDC) yesterday published its COVID-19 vaccine procurement contract with Taiwan-based Medigen Vaccine Biologics Corp (高端疫苗) after the company’s board of directors agreed to an early disclosure of the agreement.
The Chinese Nationalist Party (KMT) had repeatedly asked the CDC to publish the contract, saying the government might be concealing secrets from the public.
Premier Chen Chien-jen (陳建仁) last week said the government could not unilaterally disclose the contract due to a confidentiality agreement, but that it was negotiating with Medigen in hopes that it would agree to an early disclosure.

Photo: CNA
At an impromptu news conference yesterday afternoon, CDC Director-General Chuang Jen-hsiang (莊人祥) said Medigen informed the agency at 3:30pm yesterday that its board of directors had agreed to let it publish the contract.
Chuang said the CDC and Medigen had signed a procurement contract and a confidentiality agreement, ensuring that both sides do not disclose the details of the contract, which is in line with international commercial practices, and that many governments have also signed confidentiality agreements when they signed procurement contracts with vaccine producers.
The procurement contract was signed on May 28, 2021, according to the Public Construction Commission’s procurement contract template, but it lacked a commercial confidentiality agreement, so both sides had an oral contract to adhere to non-disclosure obligations, he said.
Medigen on June 8, 2021, asked to sign a supplementary confidentiality agreement to prevent leaks of ingredients, which was signed on July 19, 2021, he added.
According to the confidentiality agreement with Medigen, the confidentiality period was set at five years after the date of signing, but to bolster the public’s confidence, both sides agreed to disclose the information earlier, he said.
There is no agreement to keep the contract “sealed for 30 years” as some politicians have claimed, he said.
Confidentiality agreements for vaccine procurement are not new, as the CDC also signed a seven-year confidentiality agreement with Novartis when it purchased the influenza A (H1N1) monovalent vaccine in 2009, and agreements with different confidentiality periods were signed with AstraZeneca, Pfizer-BioNTech, and Moderna for their COVID-19 vaccines, he said.
Regarding vaccine prices, he said that when the Central Epidemic Command Center (CECC) announced in November 2020 that it planned to spend NT$11.5 billion (US$366.34 million) to purchase 15 million doses — or about NT$700 per dose — it was an estimated price that did not include cold chain logistics and other costs.
During the bargaining process from February to May 2021, Medigen offered a quote of NT$950 per dose, and the CDC negotiated the price to NT$881 apiece for single-dose vials and NT$810 for multi-dose vials, Chuang said.
The company must also the distribute the vaccine to no fewer than 200 locations, and be responsible for the vaccines from inspection to shipping, and the cost of disposal and other operating fees, he said.
The company had previously disclosed the prices of the vaccine and now that the full contract has been published with the consent of both sides, there is no breach of contract and it would not affect the CDC’s credit in purchasing vaccines in future, he said.
The 58-page vaccine procurement contract has been published on the CDC’s official Web site.

Considering that most countries issue more than five denominations of banknotes, the central bank has decided to redesign all five denominations, the bank said as it prepares for the first major overhaul of the banknotes in more than 24 years. Central bank Governor Yang Chin-lung (楊金龍) is expected to report to the Legislative Yuan today on the bank’s operations and the redesign’s progress. The bank in a report sent to the legislature ahead of today’s meeting said it had commissioned a survey on the public’s preferences. Survey results showed that NT$100 and NT$1,000 banknotes are the most commonly used, while NT$200 and NT$2,000

The Centers for Disease Control (CDC) yesterday reported the first case of a new COVID-19 subvariant — BA.3.2 — in a 10-year-old Singaporean girl who had a fever upon arrival in Taiwan and tested positive for the disease. The girl left Taiwan on March 20 and the case did not have a direct impact on the local community, it said. The WHO added the BA.3.2 strain to its list of Variants Under Monitoring in December last year, but this was the first imported case of the COVID-19 variant in Taiwan, CDC Deputy Director-General Lin Ming-cheng (林明誠) said. The girl arrived in Taiwan on

South Korea is planning to revise its controversial electronic arrival card, a step Taiwanese officials said prompted them to hold off on planned retaliatory measures, a South Korean media report said yesterday. A Yonhap News Agency report said that the South Korean Ministry of Foreign Affairs is planning to remove the “previous departure place” and “next destination” fields from its e-arrival card system. The plan, reached after interagency consultations, is under review and aims to simplify entry procedures and align the electronic form with the paper version, a South Korean ministry official said. The fields — which appeared only on the electronic form
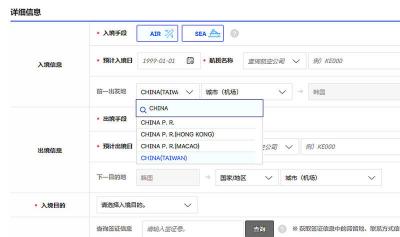
The Ministry of Foreign Affairs (MOFA) is suspending retaliation measures against South Korea that were set to take effect tomorrow, after Seoul said it is updating its e-arrival system, MOFA said today. The measures were to be a new round of retaliation after Taiwan on March 1 changed South Korea's designation on government-issued alien resident certificates held by South Korean nationals to "South Korea” from the "Republic of Korea," the country’s official name. The move came after months of protests to Seoul over its listing of Taiwan as "China (Taiwan)" in dropdown menus on its new online immigration entry system. MOFA last week