The Taiwan Foundation for Rare Disorders yesterday released its “Recommendation on the Treatment and Drug Reimbursement for Rare Diseases,” and urged the government to speed up the inclusion of rare disease treatments in the list of drugs covered by National Health Insurance (NHI).
There are about 16,000 people with rare diseases in Taiwan, which does not amount to many votes, so their rights are often neglected by presidential candidates, foundation chairperson Serena Chen (陳莉茵) said, adding that they are not protected by commercial health insurance, and need help from the NHI system the most.
Medical advances have led to more treatments for rare diseases, but they are often very expensive, and of the NHI’s global budget of about NT$809.5 billion (US$25.04 billion) last year, drugs for rare diseases only accounted for about 0.95 percent, foundation chairperson Tsai Fuu-jen (蔡輔仁) said.

Photo: Yang Yuan-ting, Taipei Times
The candidates for January’s presidential election have not proposed any policies concerning the right to healthcare of people with rare diseases, so the foundation gathered some experts to discuss solutions and propose the “Recommendation on the Treatment and Drug Reimbursement for Rare Diseases,” he said.
It takes an average of 10 to 15 years to develop a new drug for a rare disease, so up to 70 percent of rare diseases still do not have proper treatment, the foundation said.
As of last month, there were 25 types of drugs for 28 types of rare diseases waiting to be included in the NHI coverage list, which would improve 6,053 people’s health and living quality, or even save their lives, it said.
Of the 25 types of drugs, there are 12 that are the only available treatment for certain diseases, and 1,591 people are still waiting for them, the foundation said, adding that 2,020 people have died before receiving treatment covered by NHI.
Since the second-generation NHI system was launched in 2013, the average time it takes to authorize a rare disease treatment for NHI coverage has increased from 5.2 months (between 1995 and 2012) to 29.9 months, while the approval rate dropped from 86.8 percent to 52.7 percent, the foundation said.
Yang Ming-chin (楊銘欽), an adjunct professor at National Taiwan University’s Institute of Health Policy and Management, said that the longer review time and reduced approval rate affects the opportunities people have to get proper treatment.
The unspent portion of the annual budget for rare disease treatment should be reserved for the next year, Yang said.
The foundation proposed several recommendations, including increasing accessibility to drugs for treating rare diseases, committing to the UN Resolution on Addressing the Challenges of Persons Living with a Rare Disease and their Families to protect their rights, allowing reasonable growth of the NHI special budget for rare disease treatment, accelerating the review on NHI coverage of rare disease treatment, allowing patient participation in assessments and reviews and expanding financial resources to support the development of new drugs.

Considering that most countries issue more than five denominations of banknotes, the central bank has decided to redesign all five denominations, the bank said as it prepares for the first major overhaul of the banknotes in more than 24 years. Central bank Governor Yang Chin-lung (楊金龍) is expected to report to the Legislative Yuan today on the bank’s operations and the redesign’s progress. The bank in a report sent to the legislature ahead of today’s meeting said it had commissioned a survey on the public’s preferences. Survey results showed that NT$100 and NT$1,000 banknotes are the most commonly used, while NT$200 and NT$2,000

The Centers for Disease Control (CDC) yesterday reported the first case of a new COVID-19 subvariant — BA.3.2 — in a 10-year-old Singaporean girl who had a fever upon arrival in Taiwan and tested positive for the disease. The girl left Taiwan on March 20 and the case did not have a direct impact on the local community, it said. The WHO added the BA.3.2 strain to its list of Variants Under Monitoring in December last year, but this was the first imported case of the COVID-19 variant in Taiwan, CDC Deputy Director-General Lin Ming-cheng (林明誠) said. The girl arrived in Taiwan on

South Korea is planning to revise its controversial electronic arrival card, a step Taiwanese officials said prompted them to hold off on planned retaliatory measures, a South Korean media report said yesterday. A Yonhap News Agency report said that the South Korean Ministry of Foreign Affairs is planning to remove the “previous departure place” and “next destination” fields from its e-arrival card system. The plan, reached after interagency consultations, is under review and aims to simplify entry procedures and align the electronic form with the paper version, a South Korean ministry official said. The fields — which appeared only on the electronic form
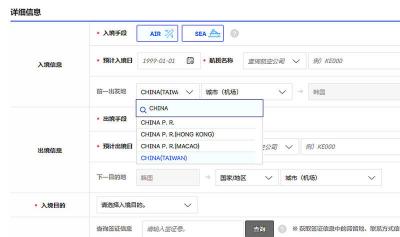
The Ministry of Foreign Affairs (MOFA) is suspending retaliation measures against South Korea that were set to take effect tomorrow, after Seoul said it is updating its e-arrival system, MOFA said today. The measures were to be a new round of retaliation after Taiwan on March 1 changed South Korea's designation on government-issued alien resident certificates held by South Korean nationals to "South Korea” from the "Republic of Korea," the country’s official name. The move came after months of protests to Seoul over its listing of Taiwan as "China (Taiwan)" in dropdown menus on its new online immigration entry system. MOFA last week