The Food and Drug Administration (FDA) yesterday released a list of 14 items that failed border inspections, including 18 batches of cherries from the US that failed border inspections in the past six months.
The 14 items included cherries from the US and Canada, truffles from Italy, chanterelle mushrooms from France, honeydew melon from Japan, cumin from China, black sesame from China, cookies from Vietnam, cardamom from India, banana paste from the Philippines and training chopsticks from China.
They failed the inspections for having excessive residue levels of pesticides, heavy metals, radioactive substances or acetic acid, and they have been returned or disposed of, the FDA said.

Photo copied by Wu Liang-yi, Taipei Times
Five items were fresh cherries — four from the US and one from Canada, the FDA said, adding that the four batches (a total of 14,160kg) from the US were found to have excessive residues of the fungicide mefentrifluconazole, while the batch (250kg) from Canada was found to have residues of the insecticide cyclaniliprole, which is banned in Taiwan.
FDA Deputy Director Lin Chin-fu (林金富) said that a total of 18 batches of cherries imported from the US have failed the border inspection for excessive mefentrifluconazole residue and therefore the sampling rate has been tightened to 100 percent.
Mefentrifluconazole can only be used on cucumbers and as Taiwan does not grow cherries, there is no maximum residue limit set for it on cherries, so if the residue concentration exceeds the limit of detection — 0.01 parts per million or more — it would fail the inspection, Lin said.
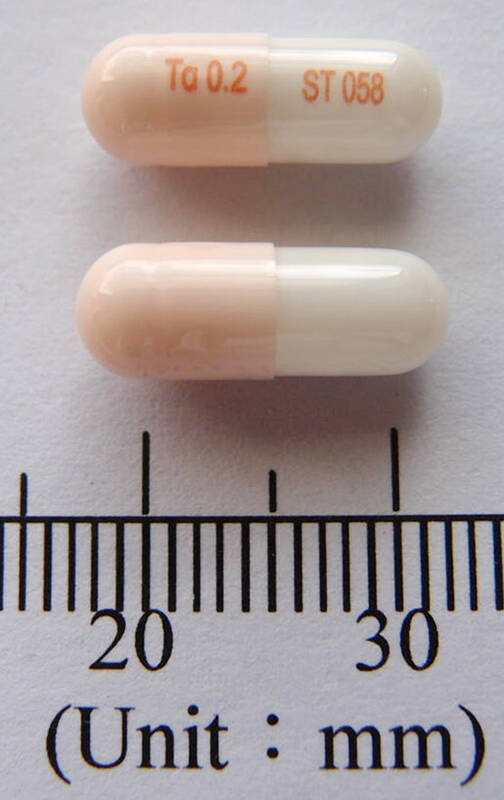
Photo courtesy of the Food and Drug Administration
Lin said the FDA has informed the American Institute in Taiwan about the situation, and recommended that it submit information and apply for a maximum residue limit for mefentrifluconazole on imported cherries so that the government could draft new regulations.
As a batch of Canadian cherries imported last year by the same importer had failed an inspection, the sampling rate was increased, he said, adding that it would be further tightened to batch-by-batch inspections until five consecutive batches pass the inspection.
Meanwhile, the FDA also issued a recall on a drug used to treat benign prostatic hyperplasia — 0.2mg Holigin S.R. Capsules manufactured by Taiwan Biotech Co Ltd (信東生技) — whose main active ingredient is tamsulosin hydrochloride.
FDA Deputy Director Chen Hwei-fang (陳惠芳) said 12 batches, about 1.7 million capsules, are being recalled as the drug has a two-year expiration date, but the company’s stability testing in the 18th month found that the dissolution performance had dropped to nearly the lowest specification limit, implying that its therapeutic effectiveness would fall below the specification before the expiration date.
The FDA said it has asked the drug company to finish recalling the drug by Aug. 24, and it also informed healthcare facilities and pharmacies to stop prescribing the drug from the 12 batches and assist in recalling the drug.
If people have concerns about the drugs they are taking they are advised to consult a doctor, it said.

Considering that most countries issue more than five denominations of banknotes, the central bank has decided to redesign all five denominations, the bank said as it prepares for the first major overhaul of the banknotes in more than 24 years. Central bank Governor Yang Chin-lung (楊金龍) is expected to report to the Legislative Yuan today on the bank’s operations and the redesign’s progress. The bank in a report sent to the legislature ahead of today’s meeting said it had commissioned a survey on the public’s preferences. Survey results showed that NT$100 and NT$1,000 banknotes are the most commonly used, while NT$200 and NT$2,000

The Centers for Disease Control (CDC) yesterday reported the first case of a new COVID-19 subvariant — BA.3.2 — in a 10-year-old Singaporean girl who had a fever upon arrival in Taiwan and tested positive for the disease. The girl left Taiwan on March 20 and the case did not have a direct impact on the local community, it said. The WHO added the BA.3.2 strain to its list of Variants Under Monitoring in December last year, but this was the first imported case of the COVID-19 variant in Taiwan, CDC Deputy Director-General Lin Ming-cheng (林明誠) said. The girl arrived in Taiwan on

ANNUAL EVENT: Two massive Pokemon balloons are to be set up in Daan Park, with an event zone operating from 10am to 6pm This year’s Taipei Floral Picnic is to be held at Daan Park today and tomorrow, featuring an exclusive Pokemon Go event, a themed food market, a coffee rave picnic area and stage performances, the Taipei Department of Information and Tourism said yesterday. Two massive Pokemon balloons are to be set up in the park as attractions, with an exclusive event zone operating from 10am to 6pm, it said. Participants who complete designated tasks on-site would have a chance to receive limited-edition souvenirs, it added. People could also try the newly launched game Pokemon Pokopia in the trial area, the department said. Three PokeStops are

South Korea is planning to revise its controversial electronic arrival card, a step Taiwanese officials said prompted them to hold off on planned retaliatory measures, a South Korean media report said yesterday. A Yonhap News Agency report said that the South Korean Ministry of Foreign Affairs is planning to remove the “previous departure place” and “next destination” fields from its e-arrival card system. The plan, reached after interagency consultations, is under review and aims to simplify entry procedures and align the electronic form with the paper version, a South Korean ministry official said. The fields — which appeared only on the electronic form