The Food and Drug Administration (FDA) has imposed restrictions on the prescription of drugs containing montelukast for people with allergic rhinitis, ruling that such medications should only be prescribed after other drugs have been used and failed to be effective.
Drugs containing montelukast could affect a person’s mental health while they are taking the medication or after treatment has stopped, with side effects including changes in behavior, unusual movements or suicidal thoughts, the FDA said in a statement yesterday.
Hung Kuo-teng (洪國登), head of the FDA’s Medicinal Products Division, told reporters that there are 31 drug permit licenses that have been issued in Taiwan for medications containing montelukast, which is used to treat asthma and allergic rhinitis, a type of nasal inflammation that occurs when the immune system overreacts to allergens in the air.
Citing National Health Insurance data, Hung said that each year, about 900,000 prescriptions of medication containing montelukast are given out.
Last year, the US Food and Drug Administration issued a warning against drugs containing montelukast, saying that their clinical effectiveness was not shown to be greater than the potential risk they posed to people’s mental health.
It then determined that they should not be used until other medications for allergic rhinitis have been used and found to be ineffective, Hung said.
In light of the warning, Taiwan’s FDA re-evaluated the effectiveness and risk of such medications, and its drug safety assessment panel determined that while drugs containing montelukast are effective in treating allergic rhinitis, they might also cause people to develop mental health problems, Hung said.
As a result, the agency has decided to list those medications as “rear-line” drugs, meaning that they can only be prescribed when other medications for nasal inflammation caused by allergies have failed to be effective, Hung said, adding that there are other drugs for treating asthma.
The ruling went into effect immediately.
The companies that hold permit licenses for drugs containing montelukast have been told to change the content of their package inserts before July 31 so that they state that the drugs can only be used when other medications for allergic rhinitis have failed to be effective, Hung said.
Companies that fail to make the change before the deadline would have their drug permit license revoked, Hung said.

Considering that most countries issue more than five denominations of banknotes, the central bank has decided to redesign all five denominations, the bank said as it prepares for the first major overhaul of the banknotes in more than 24 years. Central bank Governor Yang Chin-lung (楊金龍) is expected to report to the Legislative Yuan today on the bank’s operations and the redesign’s progress. The bank in a report sent to the legislature ahead of today’s meeting said it had commissioned a survey on the public’s preferences. Survey results showed that NT$100 and NT$1,000 banknotes are the most commonly used, while NT$200 and NT$2,000

The Centers for Disease Control (CDC) yesterday reported the first case of a new COVID-19 subvariant — BA.3.2 — in a 10-year-old Singaporean girl who had a fever upon arrival in Taiwan and tested positive for the disease. The girl left Taiwan on March 20 and the case did not have a direct impact on the local community, it said. The WHO added the BA.3.2 strain to its list of Variants Under Monitoring in December last year, but this was the first imported case of the COVID-19 variant in Taiwan, CDC Deputy Director-General Lin Ming-cheng (林明誠) said. The girl arrived in Taiwan on

South Korea is planning to revise its controversial electronic arrival card, a step Taiwanese officials said prompted them to hold off on planned retaliatory measures, a South Korean media report said yesterday. A Yonhap News Agency report said that the South Korean Ministry of Foreign Affairs is planning to remove the “previous departure place” and “next destination” fields from its e-arrival card system. The plan, reached after interagency consultations, is under review and aims to simplify entry procedures and align the electronic form with the paper version, a South Korean ministry official said. The fields — which appeared only on the electronic form
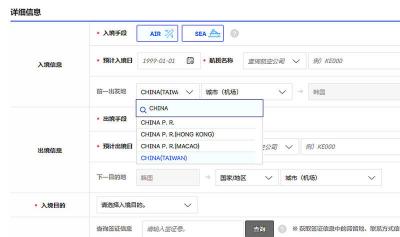
The Ministry of Foreign Affairs (MOFA) is suspending retaliation measures against South Korea that were set to take effect tomorrow, after Seoul said it is updating its e-arrival system, MOFA said today. The measures were to be a new round of retaliation after Taiwan on March 1 changed South Korea's designation on government-issued alien resident certificates held by South Korean nationals to "South Korea” from the "Republic of Korea," the country’s official name. The move came after months of protests to Seoul over its listing of Taiwan as "China (Taiwan)" in dropdown menus on its new online immigration entry system. MOFA last week