Phase 2 clinical trial results of the Medigen Vaccine Biologics Corp’s COVID-19 vaccine on Wednesday were published on the Web site of The Lancet: Respiratory Medicine, in an early preview before publication.
The study paves the way for other nations to issue emergency use authorizations or produce the Medigen vaccine, given The Lancet’s credibility as a highly respected medical journal with a rigorous peer-review process, Medigen’s international affairs director Lien Chia-en (連加恩) said.
Lien said that the study is important as it proposes methods for converting international units for efficacy comparisons.
Photo courtesy of the Food and Drug Administration
The methods have been used for correlating the efficacy of hepatitis B and HPV vaccines, but could become a first for COVID-19, he said.
The extrapolations from phase 2 data show that the Medigen vaccine has a predicted 80 to 90 percent efficacy, he said.
The leading vaccines for the pandemic have predominantly made use of mRNA technology, while the Medigen vaccine uses protein subunit technology, he said.
The phase 2 trials were double-blind, randomized and placebo-controlled, while the enrollment of more than 3,000 study participants offered an unusually large sample size, he said.
All of the participants were Asian, except for one Caucasian, he said.
National Health Research Institutes Chairman Lin Tzou-yien (林奏延), who is the principal investigator of phase 2 trials, said the EU has already approved using correlates of immunity for approving vaccines, making it more likely that the eurozone would authorize Medigen vaccines.
Yin Yi-ling (林宜玲), a research fellow at Academia Sinica’s Institute of Biomedical Research, said the institute’s biosafety level 3 lab has been fully mobilized to test samples for both Medigen’s and United Biomedical’s COVID-19 vaccines.
The task was completed within two months with the help of automation and lab partnerships whenever possible, she said.
On July 19, the Medigen vaccine became the only locally developed vaccine to receive emergency use authorization from the Food and Drug Administration after an expert panel voted in favor of the move.
The decision was criticized by some, as Medigen has yet to complete phase 3 trials, which are normally required for determining the efficacy of a vaccine.
The administration said at the time said that it granted authorization because the neutralizing antibodies generated in Medigen recipients compared well to those inoculated with AstraZeneca’s COVID-19 vaccine, a concept known as immunobridging.
Taiwan began its rollout of the Medigen vaccine on Aug. 23.
Additional reporting by Rachel Lin, Wu Liang-yi and CNA

An essay competition jointly organized by a local writing society and a publisher affiliated with the Chinese Communist Party (CCP) might have contravened the Act Governing Relations Between the People of the Taiwan Area and the Mainland Area (臺灣地區與大陸地區人民關係條例), the Mainland Affairs Council (MAC) said on Thursday. “In this case, the partner organization is clearly an agency under the CCP’s Fujian Provincial Committee,” MAC Deputy Minister and spokesperson Liang Wen-chieh (梁文傑) said at a news briefing in Taipei. “It also involves bringing Taiwanese students to China with all-expenses-paid arrangements to attend award ceremonies and camps,” Liang said. Those two “characteristics” are typically sufficient
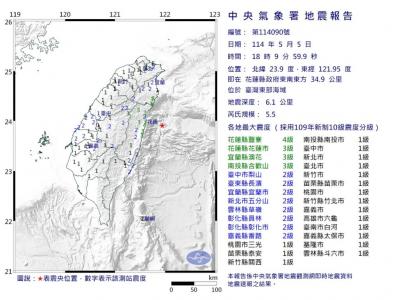
A magnitude 5.9 earthquake that struck about 33km off the coast of Hualien City was the "main shock" in a series of quakes in the area, with aftershocks expected over the next three days, the Central Weather Administration (CWA) said yesterday. Prior to the magnitude 5.9 quake shaking most of Taiwan at 6:53pm yesterday, six other earthquakes stronger than a magnitude of 4, starting with a magnitude 5.5 quake at 6:09pm, occurred in the area. CWA Seismological Center Director Wu Chien-fu (吳健富) confirmed that the quakes were all part of the same series and that the magnitude 5.5 temblor was

The Central Weather Administration has issued a heat alert for southeastern Taiwan, warning of temperatures as high as 36°C today, while alerting some coastal areas of strong winds later in the day. Kaohsiung’s Neimen District (內門) and Pingtung County’s Neipu Township (內埔) are under an orange heat alert, which warns of temperatures as high as 36°C for three consecutive days, the CWA said, citing southwest winds. The heat would also extend to Tainan’s Nansi (楠西) and Yujing (玉井) districts, as well as Pingtung’s Gaoshu (高樹), Yanpu (鹽埔) and Majia (瑪家) townships, it said, forecasting highs of up to 36°C in those areas

The brilliant blue waters, thick foliage and bucolic atmosphere on this seemingly idyllic archipelago deep in the Pacific Ocean belie the key role it now plays in a titanic geopolitical struggle. Palau is again on the front line as China, and the US and its allies prepare their forces in an intensifying contest for control over the Asia-Pacific region. The democratic nation of just 17,000 people hosts US-controlled airstrips and soon-to-be-completed radar installations that the US military describes as “critical” to monitoring vast swathes of water and airspace. It is also a key piece of the second island chain, a string of