A report by experts released by the Food and Drug Administration (FDA) on Monday raised doubts as to whether the vaccine developed by United Biomedical Inc (UBI) Asia offers sufficient protection against COVID-19 variants.
According to the report, a panel of 22 experts at a review meeting on Aug. 15, which denied a request from UBI for emergency use authorization (EUA) for its vaccine candidate UB-612, suggested that the company should conduct a third phase of clinical trials to determine the jab’s efficacy.
At the meeting, 17 of the experts, not counting the chairman, voted against UBI’s request for an EUA, as the vaccine candidate failed to meet approval standards, while the remaining four asked the company to submit more documents for another review.
For a vaccine to be granted an EUA by the FDA, it has to have a seroconversion rate of more than 50 percent and virus neutralizing antibodies — expressed as geometric mean titers (GMTs) — at least equal to two-thirds the level found in recipients of the AstraZeneca vaccine.
UBI has said that its vaccine candidate is expected to protect those inoculated against the Delta variant of SARS-CoV-2, which is now the most transmittable and contagious variant worldwide.
However, the report said that the GMT of UBI’s vaccine candidate declined at a moderate pace when faced with virus variants and based on the limited information available it is hard to say whether people who receive the vaccine would be protected against the Delta and Alpha variants.
The report also cited some experts on the panel as saying that UBI’s vaccine did not even appear to be effective against the original SARS-CoV-2 virus.
However, some other experts on the panel said that peptides inside the vaccine still have the potential to create protection against variants of SARS-CoV-2, suggesting that UBI should conduct a third phase 3 clinical trial.
They added that it remains to be seen how the vaccine creates a response to T-cells.
On Aug. 22, UBI founder Wang Chang-yi (王長怡) said that the FDA’s standards for granting EUAs were hastily designed and outdated.
She said that it would be deeply regrettable to deny people access to such a good vaccine because of hastily devised EUA standards.
Despite its application being rejected in Taiwan, UBI remains committed to seeking international approval for its COVID-19 vaccine candidate, Wang said.
The company plans to conduct phase 3 clinical trials in India for its “next generation” COVID-19 vaccine, named “UB-613,” she said.
The FDA decided to test antibody concentrations against people who have been immunized with the AstraZeneca vaccine because there is no uniform, globally accepted method of measuring GMTs or how high the GMT level should be to indicate protection.

The Centers for Disease Control (CDC) yesterday reported the first case of a new COVID-19 subvariant — BA.3.2 — in a 10-year-old Singaporean girl who had a fever upon arrival in Taiwan and tested positive for the disease. The girl left Taiwan on March 20 and the case did not have a direct impact on the local community, it said. The WHO added the BA.3.2 strain to its list of Variants Under Monitoring in December last year, but this was the first imported case of the COVID-19 variant in Taiwan, CDC Deputy Director-General Lin Ming-cheng (林明誠) said. The girl arrived in Taiwan on

South Korea is planning to revise its controversial electronic arrival card, a step Taiwanese officials said prompted them to hold off on planned retaliatory measures, a South Korean media report said yesterday. A Yonhap News Agency report said that the South Korean Ministry of Foreign Affairs is planning to remove the “previous departure place” and “next destination” fields from its e-arrival card system. The plan, reached after interagency consultations, is under review and aims to simplify entry procedures and align the electronic form with the paper version, a South Korean ministry official said. The fields — which appeared only on the electronic form
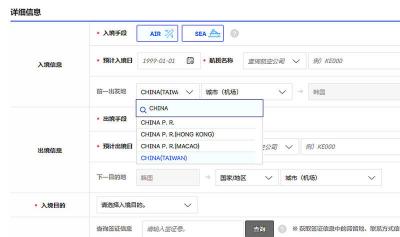
The Ministry of Foreign Affairs (MOFA) is suspending retaliation measures against South Korea that were set to take effect tomorrow, after Seoul said it is updating its e-arrival system, MOFA said today. The measures were to be a new round of retaliation after Taiwan on March 1 changed South Korea's designation on government-issued alien resident certificates held by South Korean nationals to "South Korea” from the "Republic of Korea," the country’s official name. The move came after months of protests to Seoul over its listing of Taiwan as "China (Taiwan)" in dropdown menus on its new online immigration entry system. MOFA last week

A bipartisan group of US senators has introduced a bill to enhance cooperation with Taiwan on drone development and to reduce reliance on supply chains linked to China. The proposed Blue Skies for Taiwan Act of 2026 was introduced by Republican US senators Ted Cruz and John Curtis, and Democratic US senators Jeff Merkley and Andy Kim. The legislation seeks to ease constraints on Taiwan-US cooperation in uncrewed aerial systems (UAS), including dependence on China-sourced components, limited access to capital and regulatory barriers under US export controls, a news release issued by Cruz on Wednesday said. The bill would establish a "Blue UAS