Immunobridging remains a feasible way of determining vaccine efficacy, even if the US does not yet have an official stance on the matter, the Ministry of Health and Welfare said yesterday.
The Food and Drug Administration (FDA) on Thursday last week said it would adopt the alternative approach in lieu of phase 3 trial data when evaluating emergency use authorization (EUA) for locally developed COVID-19 vaccines.
Immunobridging compares the efficacy of an approved vaccine with a trial one by measuring the concentration of neutralizing antibodies in each group of recipients.

Photo courtesy of the Executive Yuan
For a domestic vaccine to be approved, those who received the vaccine in phase 2 trials must present an antibody concentration at least as high as that found in 200 Taiwanese who have received both doses of the AstraZeneca vaccine.
Immunobridging was proposed by the WHO earlier this year and is supported by the EU, South Korea and other nations, Minister of Health and Welfare Chen Shih-chung (陳時中) said last week, although he also acknowledged that no other countries have issued this type of EUA.
However, local media have questioned international acceptance of the method, citing a statement from the US FDA.
The US regulator made the comment in response to an inquiry sent on May 20 by the Center for Drug Evaluation about the feasibility of immunobridging replacing phase 3 trials, the Chinese-language United Daily News reported.
In the message, the US FDA said an official position is not yet available, as it is “still discussing approaches to immunobridging,” the newspaper said, calling it a “slap in the face” and clear reproach of the method.
Centers for Disease Control (CDC) Deputy Director-General Chuang Jen-hsiang (莊人祥) yesterday confirmed that the CDC sent messages to US, Japanese, EU and other health authorities seeking their opinions on immunobridging.
However, the current controversy is only a matter of differing interpretations, he told reporters at the daily COVID-19 pandemic briefing.
The US regulator was unable to respond because it has not yet reached a clear conclusion, he said.
Washington was also relatively conservative about the method during a WHO meeting on May 26, but the UK, South Korea and other nations said they would accept it, while the EU voiced conditional approval, Chuang added.
Deputy Minister of Health and Welfare Hsueh Jui-yuan (薛瑞元) also weighed in, telling reporters after an Executive Yuan meeting that Washington cannot have an official stance because it is still discussing the matter.
However, this does not negate the method’s feasibility, Hsueh added.
Additional reporting by CNA

INCREASED CAPACITY: The flights on Mondays, Wednesdays, Fridays and Sundays would leave Singapore in the morning and Taipei in the afternoon Singapore Airlines is adding four supplementary flights to Taipei per week until May to meet increased tourist and business travel demand, the carrier said on Friday. The addition would raise the number of weekly flights it operates to Taipei to 18, Singapore Airlines Taiwan general manager Timothy Ouyang (歐陽漢源) said. The airline has recorded a steady rise in tourist and business travel to and from Taipei, and aims to provide more flexible travel arrangements for passengers, said Ouyang, who assumed the post in July last year. From now until Saturday next week, four additional flights would depart from Singapore on Monday, Wednesday, Friday
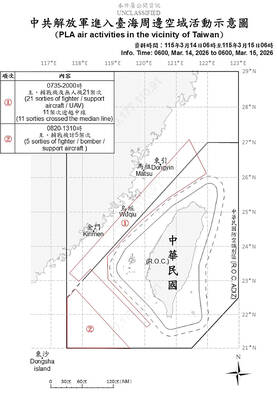
The Ministry of National Defense yesterday reported the return of large-scale Chinese air force activities after their unexplained absence for more than two weeks, which had prompted speculation regarding Beijing’s motives. China usually sends fighter jets, drones and other military aircraft around the nation on a daily basis. Interruptions to such routine are generally caused by bad weather. The Ministry of National Defense said it had detected 26 Chinese military aircraft in the Taiwan Strait over the previous 24 hours. It last reported that many aircraft on Feb. 25, when it spotted 30 aircraft, saying Beijing was carrying out another “joint combat

Taiwan successfully defended its women’s 540 kilogram title and won its first-ever men’s 640 kg title at the 2026 World Indoor Tug of War Championships in Taipei yesterday. In the women’s event, Taiwan’s eight-person squad reached the final following a round-robin preliminary round and semifinals featuring teams from Ukraine, Japan, Thailand, Vietnam, the Basque Country and South Korea. In the finals, they swept the Basque team 2-0, giving the team composed mainly of National Taiwan Normal University students and graduates its second championship in a row, and its fourth in five years. Team captain

When Paraguayan opposition lawmaker Leidy Galeano returned from an all-expenses-paid tour of six Chinese cities late last year, she was convinced Paraguay risked missing out on major economic gains by sticking with longtime ally Taipei over Beijing — a message that participants on the trip heard repeatedly from Chinese officials. “Everything I saw there, I wanted for my country,” said Galeano, a member of the newly-formed Yo Creo party whose senior figures have spoken favorably about China. This trip and others like it — which people familiar with the visits said were at the invitation of the Chinese consulate in Sao Paulo