The Food and Drug Administration (FDA) yesterday added two pharmaceutical companies to the ongoing scare centered on Taichung-based Yi Hsing Trading Co (誼興貿易), after they allegedly acquired magnesium carbonate intended for industrial use from the firm to use in the manufacture of stomach medicine.
The agency said the two firms — Jen Chang Sheng Chemical and Pharmaceutical Co (正長生化學製藥) and Mu Tsu Pharmaceutical Chemical Co (木村製藥) — are in Changhua County.
They allegedly used the substance in stomach powder products, including Jen Chang Sheng’s “Jen Chang Sheng Pulvis Stomachicus” (正長生健胃散) with marketing authorization number 003257 and Mu Tsu’s “Stomach Powder” (胃散) with authorization number 024680.
The FDA announcement came one day after three seasoning powder manufacturers in Changhua County were found to have bought magnesium carbonate meant for industrial use from Yi Hsing, using it to produce 39 products.
“Mu Tsu ceased operations on Dec. 9 last year ahead of the FDA’s new policy, implemented on Jan. 1, requiring all pharmaceutical firms to improve the quality of their medical manufacturing in line with the international PIC/S GMP standards [Pharmaceutical Inspection Convention and Pharmaceutical Inspection Cooperation Scheme],” FDA Division of Risk Management Director Lee Ming-hsin (李明鑫) said.
Companies that fail to do so will be barred from manufacturing and selling drugs and face suspension of their licenses, Lee added.
He said Mu Tsu transferred its drug licenses to another pharmaceutical firm and entrusted that company with the manufacturing of its medical products, adding that the FDA approved the transfer only last week and that no new products have been manufactured so far.
Jen Chang Sheng is currently under suspension, pending improvement of its manufacturing standards, Lee said.
“However, it is believed that there are still 10 batches of potentially tainted Jen Chang Sheng stomach powder, amounting to 900kg, circulating in the market. The last batch was produced on Dec. 8 last year. The drugs have an expiry date in 2018,” he said.
FDA Division of Medicinal Products Director Liu Li-ling (劉麗玲) said while magnesium carbonate is generally used as an excipient — or the substance that is the vehicle or medium for a drug or other active substance — in stomach powders, it must conform to the requirements of medicine ingredients or the manufacturer could be fined up to NT$300,000 and face a prison term of up to a year.
Liu said the FDA has instructed local health bureaus nationwide to recall the potentially tainted products within a month.

Considering that most countries issue more than five denominations of banknotes, the central bank has decided to redesign all five denominations, the bank said as it prepares for the first major overhaul of the banknotes in more than 24 years. Central bank Governor Yang Chin-lung (楊金龍) is expected to report to the Legislative Yuan today on the bank’s operations and the redesign’s progress. The bank in a report sent to the legislature ahead of today’s meeting said it had commissioned a survey on the public’s preferences. Survey results showed that NT$100 and NT$1,000 banknotes are the most commonly used, while NT$200 and NT$2,000

The Centers for Disease Control (CDC) yesterday reported the first case of a new COVID-19 subvariant — BA.3.2 — in a 10-year-old Singaporean girl who had a fever upon arrival in Taiwan and tested positive for the disease. The girl left Taiwan on March 20 and the case did not have a direct impact on the local community, it said. The WHO added the BA.3.2 strain to its list of Variants Under Monitoring in December last year, but this was the first imported case of the COVID-19 variant in Taiwan, CDC Deputy Director-General Lin Ming-cheng (林明誠) said. The girl arrived in Taiwan on

South Korea is planning to revise its controversial electronic arrival card, a step Taiwanese officials said prompted them to hold off on planned retaliatory measures, a South Korean media report said yesterday. A Yonhap News Agency report said that the South Korean Ministry of Foreign Affairs is planning to remove the “previous departure place” and “next destination” fields from its e-arrival card system. The plan, reached after interagency consultations, is under review and aims to simplify entry procedures and align the electronic form with the paper version, a South Korean ministry official said. The fields — which appeared only on the electronic form
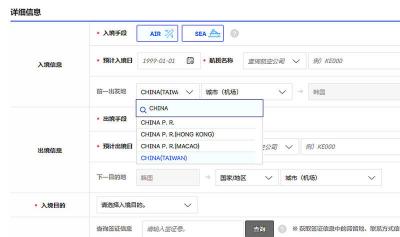
The Ministry of Foreign Affairs (MOFA) is suspending retaliation measures against South Korea that were set to take effect tomorrow, after Seoul said it is updating its e-arrival system, MOFA said today. The measures were to be a new round of retaliation after Taiwan on March 1 changed South Korea's designation on government-issued alien resident certificates held by South Korean nationals to "South Korea” from the "Republic of Korea," the country’s official name. The move came after months of protests to Seoul over its listing of Taiwan as "China (Taiwan)" in dropdown menus on its new online immigration entry system. MOFA last week