The Food and Drug Administration (FDA) on Friday ordered the recall of 38 stomach medications after a popular heartburn medication was reportedly found to contain trace amounts of a chemical that can cause cancer.
The FDA announced the recall at 7pm after receiving an alert a week ago from regulators in the US and Europe that trace amounts of the carcinogen n-nitrosodimethylamine (NDMA) was found in Zantac, a popular heartburn medication produced by British pharmaceutical company GlaxoSmithKline.
The level of NDMA was very low, so the medication was not recalled from international markets, the FDA said in a statement.
Screen grab from the Food and Drug Administration’s Web site
On receiving the alert, the administration said it launched a comprehensive inspection of all medications in Taiwan that contain ranitidine, the main ingredient in Zantac.
Twenty-one drug companies held licenses to produce 38 stomach acid drugs containing ranitidine as the main ingredient, including Culcer’s 300mg film-coated daily tablet, it said.
The FDA said that it then ordered a precautionary recall of the 38 medications.
The pharmaceutical companies must complete their recalls by tomorrow, FDA official Wu Ming-mei (吳明美) said.
They will not be allowed to market the medications until after the drugs pass a test proving that the amount of NDMA in them is within a safe range, she said.
Manufacturers failing to remove their medications from stores would face a fine of NT$60,000 to NT$300,000, according to the Consumer Protection Act (消費者保護法), Wu added.
More than 80 million tablets of stomach acid drugs containing ranitidine are prescribed to National Health Insurance patients each year, the FDA said.

FUKUOKA SITUATION: Japanese media reported that the pathogen is expected to be identified by the summer, while the CDC downplayed the idea that it was hMPV A “mysterious cold-like illness” reported in Japan’s Fukuoka Prefecture does not seem to be a new disease, but Japanese authorities have been asked about the situation, the Centers for Disease Control (CDC) said yesterday. The Fukuoka Prefectural Medical Association on Wednesday told a news conference that a “mystery cold” that has become a hot topic on social media is “highly likely to be caused by some kind of viral infection,” Japan’s KBC News reported. “Many people are experiencing symptoms starting with a sore throat, followed by a runny nose, phlegm and a severe cough,” KBC News reported, citing association officials. Health authorities are

Nvidia Corp CEO Jensen Huang (黃仁勳) arrived in Taiwan yesterday ahead of upcoming AI and technology events, saying he plans to meet with clients and Taiwan Semiconductor Manufacturing Co Chairman C.C. Wei (魏哲家) during his visit. After landing at Taipei Songshan Airport, Huang posed for photos with fans and handed out Yakult drinks to reporters and supporters waiting at the scene, saying he has “a lot to do” during the trip. Asked about reports that Nvidia’s planned headquarters site in Taipei’s Beitou Shilin Technology Park could break ground on May 27, Huang said that if the company holds an event, he would
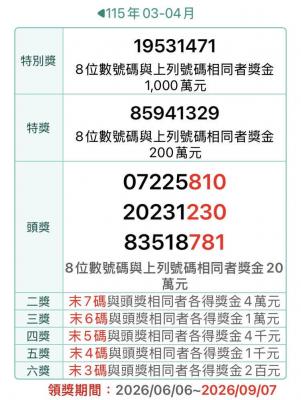
The Ministry of Finance this afternoon announced the winning numbers for the March-April uniform invoice lottery. The winning number for the NT$10 million (US$318,060) special prize is 19531471, and the winning number for the NT$2 million grand prize is 85941329. Three numbers were drawn for the NT$200,000 first prize: 07225810, 20231230 and 83518781. Those with receipts matching the last seven digits of any of the first-prize numbers will win the NT$40,000 second prize, while those matching the last six digits will win the NT$10,000 third prize. Those whose receipts match the last five digits of the first-prize numbers can claim the NT$4,000 fourth prize,

Former president Ma Ying-jeou (馬英九) yesterday expressed “shock and regret” over a statement issued by his wife without his consent, and rejected her assertion that his eldest sister, Ma Yi-nan (馬以南), would manage his medical care or act as his spokesperson amid an ongoing embezzlement probe into his foundation. Ma Ying-jeou in a statement said he was shocked by a statement by his wife, Chou Mei-ching (周美青), regarding his affairs. Ma Yi-nan “must not” be in charge of his healthcare and other personal affairs, nor can she be allowed to represent the Ma family, the former president said. He said he was also