The Food and Drug Administration (FDA) is concealing pharmaceutical companies’ violations of the law that have made members of the public “guinea pigs,” a health insurance watchdog group said yesterday.
Good Manufacturing Practice (GMP) inspections conducted by the FDA earlier this year of pharmaceutical manufacturers found that many drugs had been manufactured in a way that failed to comply with what had been described in their marketing authorizations, Democratic Progressive Party Legislator Chen Chieh-ju (陳節如) said.
The inconsistencies are in violation of the Pharmaceutical Affairs Act (藥事法), the Pharmaceutical Good Manufacturing Practice Regulations (藥物優良製造準則) and the Regulations for Registration of Medicinal Products (藥品查驗登記審查準則), and the firms and the FDA have placed patients at risk due to inadequate safety and quality of the drugs produced, Chen said in a joint statement issued with the National Health Insurance Civilian Surveillance Alliance.
The alleged changes, without seeking the approval of the regulator, have been made to package inserts, packaging and the excipient used, the group said.
However, the most dangerous of the violations has been the failure to provide bioequivalence (BE) test reports, or reports on clinical trials and whether, by monitoring the differences in blood levels, a potential to-be-marketed generic drug product is bioequivalent to a reference brand-name pharmaceutical, it added.
“As of this March, the FDA found at least 3,841 items that failed to comply with the regulations. We have no idea how many of these drugs did not have BE reports, since a request to the FDA for the relevant information has been denied by the authority, which cited the Personal Information Protection Act,” Huang Sue-ying (黃淑英), convener of the alliance, told a press conference at the legislature in Taipei.
The FDA has not asked firms producing these substandard products to suspend manufacturing nor have the products been withdrawn from the market. The list of products and the relevant information has not been made public, which means patients and healthcare providers cannot avoid using these drugs, she added.
What has enraged the group is how the drug companies’ actions were exposed. The FDA made an announcement on March 28 saying that for drugs without a BE report, but which have been on the market for at least five years and thereby have a long-term clinical experience, “their clinical experience can substitute for the required BE test if no serious adverse effects or therapeutic inequivalence have been reported.”
“This is no different to exploiting patients, allowing them to become the manufacturers’ guinea pigs at the NHI’s expense,” alliance spokeswoman Eva Teng (滕西華) said.
What is more, drugs that were manufactured and marketed after Jan. 1, 2009, and which therefore do not meet the five-year requirement, have been granted a two-year probationary period to complete BE tests without a suspension of sales of the drug during the testing period, Huang said.
“The BE test reports done by the these manufacturers are only subject to random spot-checks by the FDA,” Huang said.
In response, FDA official Tzou Meir-chyun (鄒玫君), who was also present at the press conference, said all pharmaceutical products currently on the market have been approved by the FDA, with BE tests conducteds before being approved.
“They are all post-approval changes. As post-approval changes can be, according to international standards, categorized into major and minor changes, it is only when major changes take place that a further risk assessment is needed,” Tzou said.
The FDA released a statement later in the day saying that a risk assessment meeting evaluating the drugs that underwent major changes — about 5 percent of the drugs where post-approval changes have been made — would be held within the next month.

NATIONAL SECURITY: Authorities are working to confirm the identities of the military personnel involved and investigating possible illegal conduct and regulatory violations Authorities are probing possible national security implications after Kinmen police and immigration officers on Sunday found a Chinese woman allegedly posing as a tourist while engaging in prostitution involving more than 10 military personnel. The woman, surnamed Chen (陳), has since been deported, authorities said, adding that investigators are still working to confirm the identities of those implicated, as the records only listed code names and aliases. The case stemmed from a report received by the Kinmen District Prosecutors’ Office on Friday last week from the Jinhu Precinct of the Kinmen County Police Bureau. On Sunday, police, along with the National Immigration
GLOBALGIVING: ‘ Caving to external pressure is not acceptable for an organization that has cultivated justice reform and human rights for 30 years,’ one NGO said A slew of non-government organizations (NGOs) have withdrawn from the GlobalGiving fundraising platform after it announced it would use “Chinese Taipei” instead of “Taiwan” from next month. The Taiwan Good Rice Association wrote on Facebook on Friday that it was informed on April 28 via a teleconference call of the change, which was made because the platform wanted to operate in China. Taiwan Good Rice is to terminate all cooperative relationships with GlobalGiving in response to the platform’s “unilateral and non-negotiable” decision to remove references to Taiwan, the NGO said. “Taiwan is in the official name of Taiwan Good Rice Association and the

HEAVY WEATHER: Typhoon Jangmi is due to crash straight into the Ryukyus as airlines look to shift flights to larger aircraft or cancel flights to Okinawa entirely Taiwan’s international air carriers announced flight adjustments over the weekend as Typhoon Jangmi is forecast to hit the Ryukyu Islands today and tomorrow. The Central Weather Administration (CWA) upgraded Jangmi from a tropical storm to a typhoon at 8am yesterday, with the eye located 580km south of Naha city. It was moving north at 19kph. Today, China Airlines’ CI-120, CI-121, CI-122 and CI-123 flights between Taoyuan and Naha, Okinawa, have been canceled as well as CI-132 and CI-133 between Kaohsiung and Naha. EVA Air’s BR-112, BR-113, BR-186 and BR-185 flights between Taoyuan and Naha are also canceled. Low-cost carrier Tigerair Taiwan canceled IT-230,
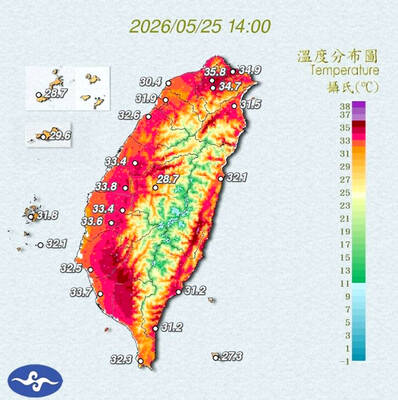
STAY COOL: The HPA recommended that people stay hydrated, use air-conditioning or fans while indoors, wear loose-fitting clothes and walk in the shade while outdoors Employers must implement measures such as installing cooling equipment, and providing drinking water and rest breaks for outdoor workers starting from Monday next week, the Taipei Department of Labor said on Sunday. Employers who fail to comply could face fines of NT$30,000 to NT$300,000 under the Occupational Safety and Health Act (職業安全衛生法), the department said. Businesses in Taipei employing fewer than 100 workers, as well as registered self-employed workers with labor insurance coverage, could receive on-site assessments and guidance from occupational safety consultants to help them apply for central government subsidies to implement or improve heat-protection measures, it said. Under the Ministry of