With close to 10,000 new cases of influenza being reported per week over the past few weeks, the Department of Health (DOH) said it would soon allow for the expanded use of the antiviral drug Tamiflu, an official said yesterday.
Since Aug. 15, anyone testing positive for the swine flu virus using a preliminary test kit has received a course of Tamiflu treatment covered by National Health Insurance, said Kuo Hsu-sung (郭旭崧), director-general of the Centers for Disease Control (CDC).
While the kits are paid for by the CDC, Taiwan is facing a shortage, Kuo said.
Those who test negative for A(H1N1) virus but who have been diagnosed with flu and have experienced exacerbations or pneumonia can obtain a prescription for Tamiflu, but will have to pay for the drug, Kuo said, adding that these cases would also be covered by National Health Insurance in the near future.
Tamiflu should not be given to all patients with flu-like symptoms without undergoing screening because that could cause drug misuse and abuse, while about 30 percent of people who use Tamiflu experience harmful side effects that are actually worse than the symptoms caused by the virus, Kuo said.
At present, hospitals and medical centers equipped with both Tamiflu and quick-test kits are mostly larger facilities, requiring patients with flu-like symptoms attending smaller hospitals or clinics to be referred to bigger institutions for testing and treatment.
The preliminary test kits, which cost between NT$250 and NT$300 each, are in short supply because of insufficient production and high production costs, Kuo said. The department would coordinate with biomedical companies to speed up production of the test kits to meet an anticipated increase in demand during the peak of the seasonal flu period next month.
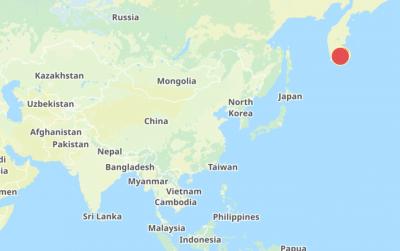
The Central Weather Administration (CWA) today issued a "tsunami watch" alert after a magnitude 8.7 earthquake struck off the Kamchatka Peninsula in northeastern Russia earlier in the morning. The quake struck off the east coast of the Kamchatka Peninsula at 7:25am (Taiwan time) at a depth of about 19km, the CWA said, citing figures from the Pacific Tsunami Warning Center. The CWA's Seismological Center said preliminary assessments indicate that a tsunami could reach Taiwan's coastal areas by 1:18pm today. The CWA urged residents along the coast to stay alert and take necessary precautions as waves as high as 1m could hit the southeastern

The National Museum of Taiwan Literature is next month to hold an exhibition in Osaka, Japan, showcasing the rich and unique history of Taiwanese folklore and literature. The exhibition, which is to run from Aug. 10 to Aug. 20 at the city’s Central Public Hall, is part of the “We Taiwan” at Expo 2025 series, highlighting Taiwan’s cultural ties with the international community, National Museum of Taiwan Literature director Chen Ying-fang (陳瑩芳) said. Folklore and literature, among Taiwan’s richest cultural heritages, naturally deserve a central place in the global dialogue, Chen said. Taiwan’s folklore would be immediately apparent at the entrance of the

Speeding and badly maintained roads were the main causes of a school bus accident on a rainy day in Taipei last year that severely injured two people and left 22 with minor injuries, the Taiwan Transportation and Safety Board said. On March 11 last year, a Kang Chiao International School bus overturned inside the Wenshan Tunnel (文山隧道) on the northbound lane of the Xinyi Expressway. The tour bus, owned by Long Lai Co, exceeded the speed limit after entering the tunnel, the board’s investigation found. Sensing that the rear of the vehicle was swaying, the driver attempted to use the service and exhaust
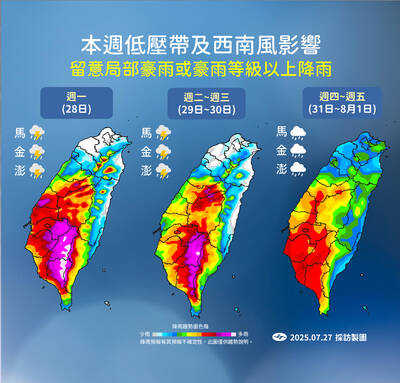
Central and southern Taiwan are to see increasingly heavy rainfall from last night through Friday due to the effects of a low-pressure system and southwesterly winds, the Central Weather Administration (CWA) said. CWA forecaster Liu Pei-teng (劉沛滕) said Tropical Storm Co-May had weakened into a low-pressure system on Saturday, but that it strengthened again into a tropical depression (TD 11) near the seas around Japan's Ryukyu Islands due to favorable environmental conditions. The tropical depression is expected to persist for two to three days, moving west-northwest by this afternoon and reaching China's Zhejiang through the East China Sea tomorrow,