For years, evidence mounted that the pain reliever Vioxx might increase the risk of heart attacks. For years, its maker, Merck, disputed such findings.
A week ago Thursday, Merck's defense started crumbling, with the arrival of irrefutable evidence from one of the company's own studies that Vioxx doubled a long-term patient's chance of having a heart attack. And Thursday, after a frantic week of internal huddles and meetings with regulators, Merck announced that it would pull the drug from the worldwide market.
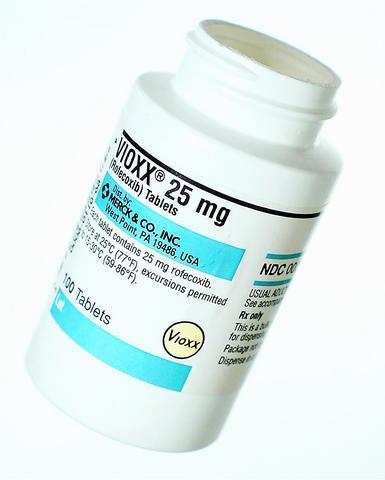
PHOTO: NY TIMES
In many ways, the short but highly profitable history of Vioxx may prove to be a story about the triumph of marketing over science. Even though worrisome evidence began to emerge shortly after the drug's approval five years ago, sales of Vioxx soared to US$2.5 billion last year on the strength of one of the biggest direct-to-consumer marketing campaigns yet for a prescription medication. In the first six months of this year alone, Merck spent an estimated US$45 million advertising the drug.
Thursday, some researchers who have long studied the drug said they were surprised, not that Vioxx was being withdrawn but that it had taken so long for the drug's death knell to be sounded.
"It is a terrifying testimony to the power of marketing," said Dr. Jerry Avorn, a divisional research director at Brigham and Women's Hospital in Boston.
Signs of Vioxx's risks emerged soon after the Food and Drug Administration approved its sale in 1999 for the treatment of acute pain and chronic pain from arthritis and other problems. The drug, which is known as a COX-2 inhibitor, did not control pain better than older, cheaper drugs. But ulcers and gastrointestinal bleeding occurred less with Vioxx.
But in 2000, Merck submitted a safety study to the FDA showing that patients taking Vioxx faced a risk of heart attacks and strokes that was four to five times higher than that of patients taking naproxen, a traditional pain reliever. The authors of the study, which was financed by Merck, theorized that the results reflected naproxen's protective effect from heart problems rather than risks posed by Vioxx.
"The investigators and the company came up with a super hypothesis that naproxen was a super drug for preventing heart attacks," said Dr. Wayne Ray, the director of the division at Vanderbilt University School of Medicine that studies the use and effects of drugs.
In 2001, the FDA warned Merck that its promotional campaigns for Vioxx were minimizing the cardiovascular risk of the drug and that it was misrepresenting the results of the 2000 study. The next year, the agency required Merck to add language to the drug's label warning about an increased risk of heart attack and stroke.
By that time, however, investigators like Ray had begun focusing on the issue of Vioxx's safety and the question of whether naproxen, which is sold under the brand name Aleve, helped prevent heart attacks. In two studies published in 2002, Ray reported that naproxen did not have a significant protective cardiovascular effect and that Vioxx, when taken at higher dosages that had become commonplace, posed an increased risk of heart-related problems.
The next major scientific finding on Vioxx appeared a year later at a medical meeting where Avorn and a colleague at Brigham and Women's Hospital, Dr. Daniel H. Solomon, reported on a Merck-financed study, based on a survey of patient records. That survey found that Vioxx, even at some moderate dosages, increased cardiovascular risk.
Merck disputed the findings of the study, and the name of a company epidemiologist who had worked on it was removed from the report before it was published in a medical journal.
In those studies, researchers did not see a similar increase in risk from Celebrex, another COX-2 inhibitor, which is made by Pfizer.
In August, Kaiser Permanente, a large nonprofit health maintenance organization, said that a review of its patient records indicated that those taking Vioxx on dosages greater than 25 milligrams faced a threefold increased risk of heart attacks and cardiovascular problems. An FDA official worked on that report.
Merck officials have long said that the earlier studies, like the Kaiser one, were not definitive because they were surveys based on patient records, rather than a clinical trial in which a drug's effectiveness and side effects are measured against a placebo in real time.
But last week, Merck received bad news from researchers in just such a trial.
The test, carefully designed to show if Vioxx was more effective than a placebo in preventing potentially cancerous colon polyps, found, instead, that the drug increased the risk of heart attack and strokes.
Janet Skidmore, a spokeswoman for Merck, said Thursday that the colon cancer study was the first clinical trial to show such results and the company took immediate action upon receiving data.
But Dr. David Campen, the medical director for drug information, utilization and medical information at Kaiser, said he thought the results of the colon cancer trial were simply another brick in the mounting body of evidence against Vioxx.
"I think they made a decision that it was just too risky for them to keep marketing the medication," he said.

China has claimed a breakthrough in developing homegrown chipmaking equipment, an important step in overcoming US sanctions designed to thwart Beijing’s semiconductor goals. State-linked organizations are advised to use a new laser-based immersion lithography machine with a resolution of 65 nanometers or better, the Chinese Ministry of Industry and Information Technology (MIIT) said in an announcement this month. Although the note does not specify the supplier, the spec marks a significant step up from the previous most advanced indigenous equipment — developed by Shanghai Micro Electronics Equipment Group Co (SMEE, 上海微電子) — which stood at about 90 nanometers. MIIT’s claimed advances last

ISSUES: Gogoro has been struggling with ballooning losses and was recently embroiled in alleged subsidy fraud, using Chinese-made components instead of locally made parts Gogoro Inc (睿能創意), the nation’s biggest electric scooter maker, yesterday said that its chairman and CEO Horace Luke (陸學森) has resigned amid chronic losses and probes into the company’s alleged involvement in subsidy fraud. The board of directors nominated Reuntex Group (潤泰集團) general counsel Tamon Tseng (曾夢達) as the company’s new chairman, Gogoro said in a statement. Ruentex is Gogoro’s biggest stakeholder. Gogoro Taiwan general manager Henry Chiang (姜家煒) is to serve as acting CEO during the interim period, the statement said. Luke’s departure came as a bombshell yesterday. As a company founder, he has played a key role in pushing for the

Taiwan Semiconductor Manufacturing Co (TSMC, 台積電) has appointed Rose Castanares, executive vice president of TSMC Arizona, as president of the subsidiary, which is responsible for carrying out massive investments by the Taiwanese tech giant in the US state, the company said in a statement yesterday. Castanares will succeed Brian Harrison as president of the Arizona subsidiary on Oct. 1 after the incumbent president steps down from the position with a transfer to the Arizona CEO office to serve as an advisor to TSMC Arizona’s chairman, the statement said. According to TSMC, Harrison is scheduled to retire on Dec. 31. Castanares joined TSMC in

EUROPE ON HOLD: Among a flurry of announcements, Intel said it would postpone new factories in Germany and Poland, but remains committed to its US expansion Intel Corp chief executive officer Pat Gelsinger has landed Amazon.com Inc’s Amazon Web Services (AWS) as a customer for the company’s manufacturing business, potentially bringing work to new plants under construction in the US and boosting his efforts to turn around the embattled chipmaker. Intel and AWS are to coinvest in a custom semiconductor for artificial intelligence computing — what is known as a fabric chip — in a “multiyear, multibillion-dollar framework,” Intel said in a statement on Monday. The work would rely on Intel’s 18A process, an advanced chipmaking technology. Intel shares rose more than 8 percent in late trading after the