Medigen Vaccine Biologics Corp should provide 5 million doses of its COVID-19 vaccine this year, according to the contract it signed with the government, Minister of Health and Welfare Chen Shih-chung (陳時中) said yesterday.
As Taiwan faces a shortage of COVID-19 vaccine doses, media reports yesterday quoted Medigen as saying on Monday that it is ahead of schedule and could provide 5 million doses by the end of the year.
The Medigen vaccine — MVC-COV1901 — was granted emergency use authorization (EUA) from the Food and Drug Administration (FDA) on July 19, and on July 25 gained approval from the Advisory Committee on Immunization Practices to be included in the national vaccination program.
Photo courtesy of the Food and Drug Administration
The Central Epidemic Command Center (CECC) previously announced that the Medigen vaccine would be administered in the next round of vaccinations, which is expected to take place at the end of this month.
As of yesterday, more than 1 million people had registered their willingness to get the Medigen vaccine on the national online COVID-19 vaccination system, and about 350,000 doses have passed the FDA’s lot release testing.
Asked about Medigen’s announcement, Chen, who heads the center, yesterday said that the drugmaker is supposed to provide 5 million doses this year, according to the procurement contract, so its production should not be considered “ahead of schedule.”
However, whether its production lines can stay on schedule and its doses pass the FDA’s lot release testing would affect the timely shipment of the drugmaker’s vaccine, he said.
As of 10am yesterday, 9,228,263 vaccine doses had been administered in Taiwan, with vaccination coverage reaching 37.11 percent for people who received at least one dose, CECC data showed.
Centers for Disease Control Deputy Director-General Chuang Jen-hsiang (莊人祥), who is the CECC’s spokesperson, said that 74 adverse events following vaccinations were reported on Monday, including five deaths one to 20 days after vaccination.
Those who died were aged 38 to 62, he said, adding that a 38-year-old who died is to undergo a post-mortem examination on Friday.
Post-mortem examinations have so far been conducted after 135 deaths following vaccinations, with the cause of death in 87 of the cases linked to underlying health conditions, he added.
In related news, Chinese Nationalist Party (KMT) Chairman Johnny Chiang (江啟臣) said that he would back a referendum petition launched by doctors Wang Wen-hsin (王文心) and Su Wei-shuo (蘇偉碩) to revoke the EUA granted to Medigen’s COVID-19 vaccine.
The proposed referendum would ask people whether they agree that the government should revoke approval of COVID-19 vaccines that have not completed phase 3 clinical trials, but only completed a review involving immunobridging.
Granting the Medigen vaccine a EUA “through an improper procedure” has raised questions among members of the public, as well as opposition parties and the medical community, Chiang wrote on Facebook.
The petition seeks 100,000 joint proposers — to prevent the Central Election Commission from barring the proposal on a technicality — and hopes to garner 500,000 signatures in six months, he wrote.
Chiang said that he was willing to “join the fight” and become a joint proposer.
Additional reporting by Sherry Hsiao

Considering that most countries issue more than five denominations of banknotes, the central bank has decided to redesign all five denominations, the bank said as it prepares for the first major overhaul of the banknotes in more than 24 years. Central bank Governor Yang Chin-lung (楊金龍) is expected to report to the Legislative Yuan today on the bank’s operations and the redesign’s progress. The bank in a report sent to the legislature ahead of today’s meeting said it had commissioned a survey on the public’s preferences. Survey results showed that NT$100 and NT$1,000 banknotes are the most commonly used, while NT$200 and NT$2,000

The Centers for Disease Control (CDC) yesterday reported the first case of a new COVID-19 subvariant — BA.3.2 — in a 10-year-old Singaporean girl who had a fever upon arrival in Taiwan and tested positive for the disease. The girl left Taiwan on March 20 and the case did not have a direct impact on the local community, it said. The WHO added the BA.3.2 strain to its list of Variants Under Monitoring in December last year, but this was the first imported case of the COVID-19 variant in Taiwan, CDC Deputy Director-General Lin Ming-cheng (林明誠) said. The girl arrived in Taiwan on

South Korea is planning to revise its controversial electronic arrival card, a step Taiwanese officials said prompted them to hold off on planned retaliatory measures, a South Korean media report said yesterday. A Yonhap News Agency report said that the South Korean Ministry of Foreign Affairs is planning to remove the “previous departure place” and “next destination” fields from its e-arrival card system. The plan, reached after interagency consultations, is under review and aims to simplify entry procedures and align the electronic form with the paper version, a South Korean ministry official said. The fields — which appeared only on the electronic form
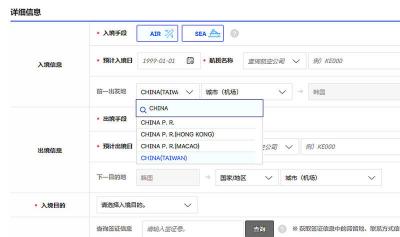
The Ministry of Foreign Affairs (MOFA) is suspending retaliation measures against South Korea that were set to take effect tomorrow, after Seoul said it is updating its e-arrival system, MOFA said today. The measures were to be a new round of retaliation after Taiwan on March 1 changed South Korea's designation on government-issued alien resident certificates held by South Korean nationals to "South Korea” from the "Republic of Korea," the country’s official name. The move came after months of protests to Seoul over its listing of Taiwan as "China (Taiwan)" in dropdown menus on its new online immigration entry system. MOFA last week