The Food and Drug Administration (FDA) yesterday said that another batch of AstraZeneca’s lipid-lowering drug Crestor was found to have been adulterated with counterfeit drugs, so the company has agreed to initiate a recall for all batches on the market.
The administration last weekend confirmed that batch number MV503 of Crestor 10mg film-coated tablets contained counterfeit drugs in similar packaging, and all the medication from that batch was recalled from hospitals and pharmacies.
“The agency received the report about the counterfeit drugs on Thursday last week and immediately launched an investigation; examinations confirmed that there were counterfeit drugs being sold,” administration Director-General Shou-Mei Wu (吳秀梅) said yesterday.

Photo: courtesy of the Taichung Health Bureau
The New Taipei District Prosecutors’ Office took two suspects into custody over the weekend, and they may face up to 10 years in prison or a fine up to NT$100 million (US$3.24 million) for producing counterfeit drugs under the Pharmaceutical Affairs Act (藥事法).
Another consignment of Crestor 10mg film-coated tablets, batch number MK479, was also found to contain counterfeit drugs, so the FDA said it asked AstraZeneca to recall all batches of the product in the interests of public safety.
“The main ingredient that has been added to the counterfeit drugs is actually another type of lipid-lowering drug, atorvastatin, that used to be commonly prescribed, so people who have taken the counterfeit drugs need not worry too much,” Wu said, adding that people should consult doctors or pharmacists if they have any questions.
According to National Health Insurance (NHI) Administration statistics, more than 200,000 boxes of Crestor are prescribed each month in Taiwan by NHI physicians, administration official Chih Lan-hui (遲蘭慧) said.
AstraZeneca told the administration that it has a sufficient quantity of the medication in stock to replace the recalled products, so patients would not be forced to take other types of drugs, she said.
Asked to respond to media reports that two other types of oral lipid-lowering drugs and one type of oral hypoglycemic drugs had been adulterated with counterfeit drugs and marketed under other brand names, the administration said it did not have any information on the issue, but that prosecutors and the administration would investigate.
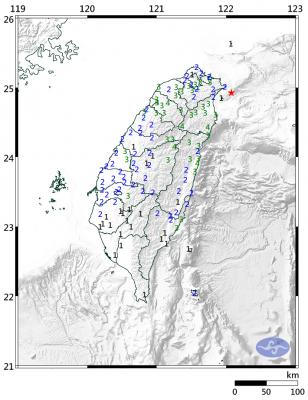
A magnitude 6.1 earthquake struck off the coast of Yilan County at 8:39pm tonight, the Central Weather Administration (CWA) said, with no immediate reports of damage or injuries. The epicenter was 38.7km east-northeast of Yilan County Hall at a focal depth of 98.3km, the CWA’s Seismological Center said. The quake’s maximum intensity, which gauges the actual physical effect of a seismic event, was a level 4 on Taiwan’s 7-tier intensity scale, the center said. That intensity level was recorded in Yilan County’s Nanao Township (南澳), Hsinchu County’s Guansi Township (關西), Nantou County’s Hehuanshan (合歡山) and Hualien County’s Yanliao (鹽寮). An intensity of 3 was

Instead of focusing solely on the threat of a full-scale military invasion, the US and its allies must prepare for a potential Chinese “quarantine” of Taiwan enforced through customs inspections, Stanford University Hoover fellow Eyck Freymann said in a Foreign Affairs article published on Wednesday. China could use various “gray zone” tactics in “reconfiguring the regional and ultimately the global economic order without a war,” said Freymann, who is also a nonresident research fellow at the US Naval War College. China might seize control of Taiwan’s links to the outside world by requiring all flights and ships entering or leaving Taiwan

The first of 10 new high-capacity trains purchased from South Korea’s Hyundai Rotem arrived at the Port of Taipei yesterday to meet the demands of an expanding metro network, Taipei Rapid Transit Corp (TRTC) said yesterday. The train completed a three-day, 1,200km voyage from the Port of Masan in South Korea, the company said. Costing NT$590 million (US$18.79 million) each, the new six-carriage trains feature a redesigned interior based on "human-centric" transportation concepts, TRTC said. The design utilizes continuous longitudinal seating to widen the aisles and optimize passenger flow, while also upgrading passenger information displays and driving control systems for a more comfortable

Taiwan's first indigenous defense submarine, the SS-711 Hai Kun (海鯤, or Narwhal), departed for its 13th sea trial at 7am today, marking its seventh submerged test, with delivery to the navy scheduled for July. The outing also marked its first sea deployment since President William Lai (賴清德) boarded the submarine for an inspection on March 19, drawing a crowd of military enthusiasts who gathered to show support. The submarine this morning departed port accompanied by CSBC Corp’s Endeavor Manta (奮進魔鬼魚號) uncrewed surface vessel and a navy M109 assault boat. Amid public interest in key milestones such as torpedo-launching operations and overnight submerged trials,