The Centers for Disease Control (CDC) yesterday issued an emergency call to stop issuing a flu vaccine in seven counties and cities after the Taipei Department of Health on Wednesday reported a discolored dose.
The suspension affects the Vaxigrip brand of trivalent flu vaccine manufactured by Lyon, France-based Sanofi Pasteur of the same lot number, the CDC said.
The government has prepared 6 million doses for this year’s free flu shot program, which hospitals began administering on Oct. 15, the centers said, adding that Vaxigrip is one of the two vaccines it uses.
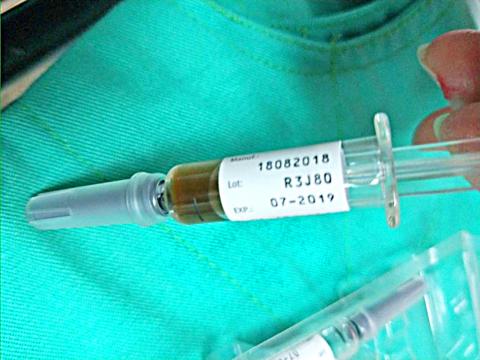
Photo courtesy of the Centers for Disease Control
The other type, AdimFlu-S, is manufactured by Taiwan-based Adimmune Corp (國光生技), it said.
A Taipei-based medical worker administering flu shots at a mobile vaccination station saw that the vaccine in one of the syringes had a brownish color, while normal vaccines are transparent, the CDC said, adding that the discolored vaccine was not administered.
CDC officials inspected the Vaxigrip stock at its storage facility and did not find any other vaccine with abnormal coloring.
The nation has imported 300,000 Vaxigrip doses, with about 190,000 syringes having already been distributed to health centers in Taipei, New Taipei City, Hsinchu city and county, as well as Taoyuan, Taichung and Miaoli County, while about 100,000 syringes were still in storage, the centers said.
As of yesterday afternoon, more than 32,000 Vaxigrip doses had been administered, it said.
CDC Deputy Director-General Chuang Jen-hsiang (莊人祥) said this was the first time that the centers had seen discoloration of an influenza vaccine.
The centers would return the discolored vaccine to Sanofi Pasteur for further analysis and all vaccines in the batch are to be placed on hold until the company has finished its probe, Chuang said.
The Food and Drug Administration said it has asked Sanofi Pasteur to report the vaccines as defective and explain the discoloration as soon as possible.
Sanofi Pasteur in a statement said that the discolored sample was being delivered to the company’s headquarters for analysis.
As there are no reports of similar incidents with the company’s other vaccines, Sanofi Pasteur said it was regarding the case as an isolated incident.

NO HUMAN ERROR: After the incident, the Coast Guard Administration said it would obtain uncrewed aerial vehicles and vessels to boost its detection capacity Authorities would improve border control to prevent unlawful entry into Taiwan’s waters and safeguard national security, the Mainland Affairs Council (MAC) said yesterday after a Chinese man reached the nation’s coast on an inflatable boat, saying he “defected to freedom.” The man was found on a rubber boat when he was about to set foot on Taiwan at the estuary of Houkeng River (後坑溪) near Taiping Borough (太平) in New Taipei City’s Linkou District (林口), authorities said. The Coast Guard Administration’s (CGA) northern branch said it received a report at 6:30am yesterday morning from the New Taipei City Fire Department about a

IN BEIJING’S FAVOR: A China Coast Guard spokesperson said that the Chinese maritime police would continue to carry out law enforcement activities in waters it claims The Philippines withdrew its coast guard vessel from a South China Sea shoal that has recently been at the center of tensions with Beijing. BRP Teresa Magbanua “was compelled to return to port” from Sabina Shoal (Xianbin Shoal, 仙濱暗沙) due to bad weather, depleted supplies and the need to evacuate personnel requiring medical care, the Philippine Coast Guard (PCG) spokesman Jay Tarriela said yesterday in a post on X. The Philippine vessel “will be in tiptop shape to resume her mission” after it has been resupplied and repaired, Philippine Executive Secretary Lucas Bersamin, who heads the nation’s maritime council, said

CHINA POLICY: At the seventh US-EU Dialogue on China, the two sides issued strong support for Taiwan and condemned China’s actions in the South China Sea The US and EU issued a joint statement on Wednesday supporting Taiwan’s international participation, notably omitting the “one China” policy in a departure from previous similar statements, following high-level talks on China and the Indo-Pacific region. The statement also urged China to show restraint in the Taiwan Strait. US Deputy Secretary of State Kurt Campbell and European External Action Service Secretary-General Stefano Sannino cochaired the seventh US-EU Dialogue on China and the sixth US-EU Indo-Pacific Consultations from Monday to Tuesday. Since the Indo-Pacific consultations were launched in 2021, references to the “one China” policy have appeared in every statement apart from the

More than 500 people on Saturday marched in New York in support of Taiwan’s entry to the UN, significantly more people than previous years. The march, coinciding with the ongoing 79th session of the UN General Assembly, comes close on the heels of growing international discourse regarding the meaning of UN Resolution 2758. Resolution 2758, adopted by the UN General Assembly in 1971, recognizes the People’s Republic of China (PRC) as the “only lawful representative of China.” It resulted in the Republic of China (ROC) losing its seat at the UN to the PRC. Taiwan has since been excluded from