Moderna Inc’s experimental vaccine was 94.5 percent effective in preventing COVID-19 based on interim data from a late-stage trial, the company said yesterday, becoming the second US drugmaker to report results that far exceed expectations.
Together with Pfizer Inc’s vaccine, which is also more than 90 percent effective, and pending more safety data and regulatory review, the US could have two vaccines authorized for emergency use next month with as many as 60 million doses of vaccine available this year.
Next year, the US government could have access to more than 1 billion doses just from the two vaccine makers, more than needed for the country’s 330 million residents.
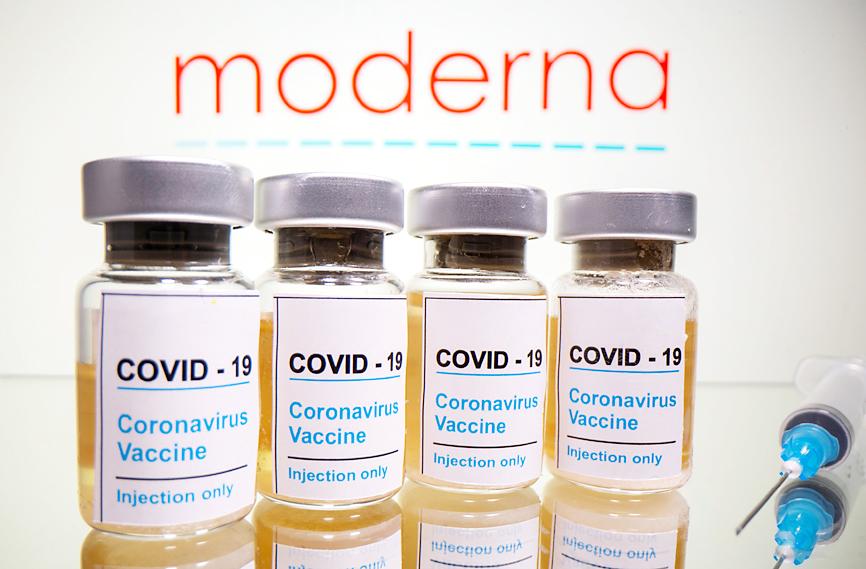
Photo: Reuters
The vaccines, both developed with new technology known as messenger RNA (mRNA), represent powerful tools to fight a pandemic that has infected 54 million people worldwide and killed 1.3 million.
The news also comes at a time when COVID-19 cases are soaring, hitting new records in the US and pushing some European countries back into lockdowns.
“We are going to have a vaccine that can stop COVID-19,” Moderna president Stephen Hoge said in a telephone interview.
Moderna’s interim analysis was based on 95 infections among trial participants who received either a placebo or the vaccine. Of those, only five infections occurred in those who received the vaccine, which is administered in two shots 28 days apart.
“This news from Moderna is tremendously exciting and considerably boosts optimism that we will have a choice of good vaccines in the next few months,” said Peter Openshaw, professor of experimental medicine at Imperial College London.
“This latest press release is based on a study of 30,000 US adults, including many high-risk or elderly persons. This gives us confidence that the results are relevant in the people who are most at risk of COVID-19,” he said.
Moderna expects to have enough safety data required for US authorization in the next week or so and the company expects to file for emergency use authorization in the coming weeks.
A key advantage of Moderna’s vaccine is that it does not need ultra-cold storage like Pfizer’s, making it easier to distribute. Moderna expects it to be stable at normal fridge temperatures of 2°C to 8°C for 30 days and it can be stored for up to six months at minus-20°C.
Pfizer’s vaccine must be shipped and stored at minus-70°C. It can be stored for up to five days at standard refrigerator temperatures or for up to 15 days in a thermal shipping box.
The data from Moderna’s trial involving 30,000 volunteers also showed that the vaccine prevented cases of severe COVID-19, a question that still remains with the Pfizer vaccine.
Moderna expects to produce about 20 million doses for the US this year, millions of which it has already made and is ready to ship if it gets US Food and Drug Administration authorization.

RETHINK? The defense ministry and Navy Command Headquarters could take over the indigenous submarine project and change its production timeline, a source said Admiral Huang Shu-kuang’s (黃曙光) resignation as head of the Indigenous Submarine Program and as a member of the National Security Council could affect the production of submarines, a source said yesterday. Huang in a statement last night said he had decided to resign due to national security concerns while expressing the hope that it would put a stop to political wrangling that only undermines the advancement of the nation’s defense capabilities. Taiwan People’s Party Legislator Vivian Huang (黃珊珊) yesterday said that the admiral, her older brother, felt it was time for him to step down and that he had completed what he
Taiwan has experienced its most significant improvement in the QS World University Rankings by Subject, data provided on Sunday by international higher education analyst Quacquarelli Symonds (QS) showed. Compared with last year’s edition of the rankings, which measure academic excellence and influence, Taiwanese universities made great improvements in the H Index metric, which evaluates research productivity and its impact, with a notable 30 percent increase overall, QS said. Taiwanese universities also made notable progress in the Citations per Paper metric, which measures the impact of research, achieving a 13 percent increase. Taiwanese universities gained 10 percent in Academic Reputation, but declined 18 percent

CHINA REACTS: The patrol and reconnaissance plane ‘transited the Taiwan Strait in international airspace,’ the 7th Fleet said, while Taipei said it saw nothing unusual The US 7th Fleet yesterday said that a US Navy P-8A Poseidon flew through the Taiwan Strait, a day after US and Chinese defense heads held their first talks since November 2022 in an effort to reduce regional tensions. The patrol and reconnaissance plane “transited the Taiwan Strait in international airspace,” the 7th Fleet said in a news release. “By operating within the Taiwan Strait in accordance with international law, the United States upholds the navigational rights and freedoms of all nations.” In a separate statement, the Ministry of National Defense said that it monitored nearby waters and airspace as the aircraft

UNDER DISCUSSION: The combatant command would integrate fast attack boat and anti-ship missile groups to defend waters closest to the coastline, a source said The military could establish a new combatant command as early as 2026, which would be tasked with defending Taiwan’s territorial waters 24 nautical miles (44.4km) from the nation’s coastline, a source familiar with the matter said yesterday. The new command, which would fall under the Naval Command Headquarters, would be led by a vice admiral and integrate existing fast attack boat and anti-ship missile groups, along with the Naval Maritime Surveillance and Reconnaissance Command, said the source, who asked to remain anonymous. It could be launched by 2026, but details are being discussed and no final timetable has been announced, the source