The pharmaceutical giant that makes a promising COVID-19 drug has registered it as a rare disease treatment with US regulators, a status that can potentially be worth millions in tax breaks and competition-free sales.
What that specialty status will actually mean for the marketing or profitability of Gilead Science Inc’s experimental drug remdesivir is not clear.
The drugmaker did not immediately respond on Tuesday to requests for comment.

Photo: Bloomberg
Experts who have studied the so-called “orphan drug” program have said that the company’s request — and the US Food and Drug Administration’s (FDA) decision to grant it — seem inappropriate given the rapidly expanding threat of the viral outbreak.
A financial analyst, though, called Gilead’s request “pretty standard.”
The FDA granted the status on Monday, according to the agency’s Web site.
If approved for coronavirus, Gilead Sciences would receive seven years of exclusive US marketing for the drug, and tax credits on its research and development costs.
The US Congress created the orphan drug program more than 35 years ago to encourage companies to develop drugs for niche diseases and conditions that might not otherwise be profitable.
However, since then, filing for orphan status has become a standard pharmaceutical industry tactic to extend the profitability of drugs and block competitors. Orphan drugs are also typically eligible for other special programs that speed up FDA reviews for approval.
The FDA defines a rare disease as one with fewer than 200,000 patients in the US.
In a statement on Tuesday, the agency said COVID-19 fit that criteria when the request was made.
There are more than 50,000 cases in the US, but many more expected in the coming weeks and months.
“It seems like a misuse of the Orphan Drug Act, even though technically it’s within the bounds of the law,” said Aaron Kesselheim, a Harvard Medical School health policy expert. “There’s no expectation here that this drug wouldn’t be able to generate appropriate revenue for the manufacturer.”
Kesselheim said a number of the early AIDS drugs also received orphan drug status in the 1980s and 1990s, but then went on to generate billions in sales.
However, Tyler Van Buren, a senior research analyst at the financial services firm Piper Sandler Cos, called Gilead’s filing “pretty standard.”
“It says nothing about profiting off of the pandemic, but it does provide protection if remdesivir turns into a business in subsequent years,” he said.
The orphan drug program has come under scrutiny from the media, Congress and government inspectors amid concerns that it is being misused to protect six-figure prices on specialty drugs.
About half of the 48 new drugs approved by the agency last year received orphan drug designation. Many were priced well above US$100,000 for a year’s supply, including drugs for cancer, muscular dystrophy and other genetic disorders.
The nonprofit Public Citizen group said in a statement that the US government should be “urgently concerned” with the affordability of remdesivir.
Gilead chairman and chief executive officer Daniel O’Day has previously said that the company has not discussed with any governments how much remdesivir will cost.
“The topic of pricing comes up once you know the medicine works,” he said.
Day earlier this month told a news conference with other drug industry executives that his company has been working on the drug’s development for a decade.

Taiwan Transport and Storage Corp (TTS, 台灣通運倉儲) yesterday unveiled its first electric tractor unit — manufactured by Volvo Trucks — in a ceremony in Taipei, and said the unit would soon be used to transport cement produced by Taiwan Cement Corp (TCC, 台灣水泥). Both TTS and TCC belong to TCC International Holdings Ltd (台泥國際集團). With the electric tractor unit, the Taipei-based cement firm would become the first in Taiwan to use electric vehicles to transport construction materials. TTS chairman Koo Kung-yi (辜公怡), Volvo Trucks vice president of sales and marketing Johan Selven, TCC president Roman Cheng (程耀輝) and Taikoo Motors Group

Stephen Garrett, a 27-year-old graduate student, always thought he would study in China, but first the country’s restrictive COVID-19 policies made it nearly impossible and now he has other concerns. The cost is one deterrent, but Garrett is more worried about restrictions on academic freedom and the personal risk of being stranded in China. He is not alone. Only about 700 American students are studying at Chinese universities, down from a peak of nearly 25,000 a decade ago, while there are nearly 300,000 Chinese students at US schools. Some young Americans are discouraged from investing their time in China by what they see
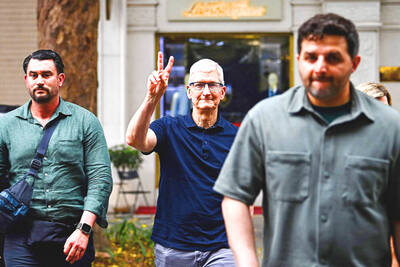
MAJOR DROP: CEO Tim Cook, who is visiting Hanoi, pledged the firm was committed to Vietnam after its smartphone shipments declined 9.6% annually in the first quarter Apple Inc yesterday said it would increase spending on suppliers in Vietnam, a key production hub, as CEO Tim Cook arrived in the country for a two-day visit. The iPhone maker announced the news in a statement on its Web site, but gave no details of how much it would spend or where the money would go. Cook is expected to meet programmers, content creators and students during his visit, online newspaper VnExpress reported. The visit comes as US President Joe Biden’s administration seeks to ramp up Vietnam’s role in the global tech supply chain to reduce the US’ dependence on China. Images on

New apartments in Taiwan’s major cities are getting smaller, while old apartments are increasingly occupied by older people, many of whom live alone, government data showed. The phenomenon has to do with sharpening unaffordable property prices and an aging population, property brokers said. Apartments with one bedroom that are two years old or older have gained a noticeable presence in the nation’s six special municipalities as well as Hsinchu county and city in the past five years, Evertrust Rehouse Co (永慶房產集團) found, citing data from the government’s real-price transaction platform. In Taipei, apartments with one bedroom accounted for 19 percent of deals last