Hou Zhihui breaks open a cold-medicine capsule, pours the powder on to a piece of steamed dough and folds it together. He passes the miniature bun to a colleague, who pops it in her mouth.
That is his response to the discovery of 77 million capsules made of industrial gel containing chromium, a carcinogenic heavy metal, the latest in a series of safety problems blighting China’s healthcare industry, including the widespread manufacture of fake drugs.
The Chinese government has repeatedly promised to tighten regulatory systems after safety scandals involving fish, drugs, toys, toothpaste, children’s clothes, tires, drugs and milk fortified with melamine, used in the manufacture of tabletops. However, little has been done apart from a few, highly publicized arrests.
“I read about the capsule problem ... the next morning, a colleague of mine had a cold, so I thought of a way for her to take the medicine,” Hou said.
China announced last month that 254 pharmaceutical suppliers, or 12.7 percent of the total, were producing tainted capsules. At least 10 are listed or linked to China-listed firms, according to official Chinese media. Of 11,561 batches of drugs tested, 5.8 percent contained excessive levels of chromium.
Instead of using gelatine derived from animal parts, they used cheap industrial gelatine from leather scraps treated with chromium that tans and softens animal hide.
The problem is pervasive because of the pressure to produce low-cost drugs and still make a profit, and the popularity of traditional medicine, which is often made into powders and packed into capsules.
Authorities swooped in on 236 capsule makers, ordered 42 of them to stop production, closed 84 production lines, revoked the licenses of seven companies and referred 13 to the police.
As China revamps its healthcare system to make it more affordable to its 1.3 billion population, bureaucrats have gone all out to lower drug prices in large drug tenders that give winners a captive market spanning several provinces.
Toxic capsules have also been uncovered in Hong Kong, where drugmakers source 90 percent of their capsules from China.
“The current tendering system of the essential drug list has three major problems: First, essential drug tendering in provinces has become a political competition to depress prices among local governments, which results in constant decreases in prices of some essential drugs,” the Chinese Pharmaceutical Industry Association said in a report.
Analysts say the cutting of corners would not go away overnight.
“It will continue to haunt the market as long as enterprises have to make a profit,” said Gideon Lo (羅偉業), analyst at Nomura Equity Research in Hong Kong. “Consolidation must happen and is already happening. It will be good if we can see the number of pharmaceutical makers coming down to 1,000 [from more than 6,000] in the next five years.”
As for breaking open capsules and taking their contents separately, experts say that is dangerous, assuming the toxic capsules work, as they should, on a timer.
“The formulation of some drugs is prepared for sustained release, meaning the drug is prepared in a way to release the drug slowly,” said William Chui (崔俊明), president of the Society of Hospital Pharmacists of Hong Kong. “If you break the capsule, you destroy the mechanism of sustained release. The drug will immediately be released and you risk increasing the drug level in your blood and developing side effects.”
Nearly half of China’s major cities are suffering “moderate to severe” levels of subsidence, putting millions of people at risk of flooding, especially as sea levels rise, according to a study of nationwide satellite data released yesterday. The authors of the paper, published by the journal Science, found that 45 percent of China’s urban land was sinking faster than 3mm per year, with 16 percent at more than 10mm per year, driven not only by declining water tables, but also the sheer weight of the built environment. With China’s urban population already in excess of 900 million people, “even a small portion
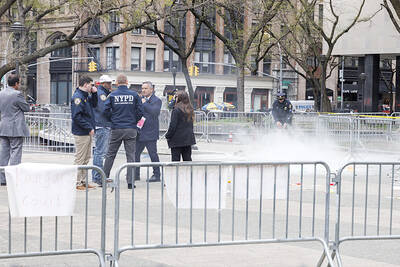
UNSETTLING IMAGES: The scene took place in front of TV crews covering the Trump trial, with a CNN anchor calling it an ‘emotional and unbelievably disturbing moment’ A man who doused himself in an accelerant and set himself on fire outside the courthouse where former US president Donald Trump is on trial has died, police said yesterday. The New York City Police Department (NYPD) said the man was declared dead by staff at an area hospital. The man was in Collect Pond Park at about 1:30pm on Friday when he took out pamphlets espousing conspiracy theories, tossed them around, then doused himself in an accelerant and set himself on fire, officials and witnesses said. A large number of police officers were nearby when it happened. Some officers and bystanders rushed

Beijing is continuing to commit genocide and crimes against humanity against Uyghurs and other Muslim minorities in its western Xinjiang province, U.S. Secretary of State Antony Blinken said in a report published on Monday, ahead of his planned visit to China this week. The State Department’s annual human rights report, which documents abuses recorded all over the world during the previous calendar year, repeated language from previous years on the treatment of Muslims in Xinjiang, but the publication raises the issue ahead of delicate talks, including on the war in Ukraine and global trade, between the top U.S. diplomat and Chinese

HYPOCRISY? The Chinese Ministry of Foreign Affairs yesterday asked whether Biden was talking about China or the US when he used the word ‘xenophobic’ US President Joe Biden on Wednesday called for a hike in steel tariffs on China, accusing Beijing of cheating as he spoke at a campaign event in Pennsylvania. Biden accused China of xenophobia, too, in a speech to union members in Pittsburgh. “They’re not competing, they’re cheating. They’re cheating and we’ve seen the damage here in America,” Biden said. Chinese steel companies “don’t need to worry about making a profit because the Chinese government is subsidizing them so heavily,” he said. Biden said he had called for the US Trade Representative to triple the tariff rates for Chinese steel and aluminum if Beijing was