Indian pharmaceutical tycoon Yusuf Hamied revolutionized AIDS treatment more than a decade ago by supplying cut-price drugs to the world’s poor — and now he wants to do the same for cancer.
Hamied, chairman of generic drugs giant Cipla, last month slashed the cost of three medicines to fight brain, kidney and lung cancer in India, making the drugs up to more than four times cheaper.
“I hope we’ll cut prices of many more cancer drugs,” he said, adding that he wants to supply the cheaper drugs to Africa and elsewhere. “Reducing the price of cancer drugs is a humanitarian move.”
Hamied, 76, was pilloried by Western drug giants 11 years ago when he broke their monopoly by offering to supply life-saving triple therapy AIDS drug cocktails for under US$1 a day — one-thirtieth the price set by the multinationals.
The firms branded him an intellectual property thief while he accused them of being “global serial killers” whose high prices were costing the lives of AIDS patients.
“What he did was path-breaking. It has been very important in saving lives, and what he is doing with cancer drugs is the same,” said Leena Menghaney, a lawyer with humanitarian group Medecins sans Frontieres (Doctors Without Borders).
In 1972, India made only the process for making drugs patentable, not the drugs themselves.
This meant firms could “reverse-engineer” or change methods used to make medicines and sell them at up to one-fiftieth of US prices.
The legislation gave a huge leg-up to India’s generics industry and gave the nation the nickname “the pharmacy to the Third World.”
However, in 2005, India brought its law in line with WTO rules recognizing 20-year patents, pushing up the prices of newly launched drugs.
Cipla, India’s fourth-largest pharmaceutical company by sales, has been pressing the government to allow widespread use of “compulsory licenses,” which are permitted under WTO rules.
The licenses allow companies to make existing life-saving drugs to sell in countries where they are otherwise priced out of reach.
India’s first such license was granted in March to Natco Pharma to produce a generic version of Bayer’s blockbuster kidney cancer drug Nexavar, cutting the price from 28,000 rupees (US$500) for a monthly dose to 6,840 rupees.
Ranjit Shahani, who heads the Organisation of Pharmaceutical Producers of India, says widespread compulsory licensing will jeopardize investment in innovative pharmaceuticals.
Bayer has said it will launch a legal challenge to the compulsory license, and global drugmakers have vowed to oppose the spread of such legislation.
Hamied denied that his latest move was simply an attempt to boost his share in the oncology drugs market, insisting business must be linked to “social responsibility.”
However, he said like any other business, his company, which has 23 cancer drugs, also wants sales.
“I owe it to my shareholders to be pragmatic,” he said.
Born in Lithuania to an Indian Muslim father and a Lithuanian Jewish mother, Hamied was two months old when his parents fled Europe in the 1930s under the threat of Nazi Germany.
He was raised in Mumbai and studied for a doctorate in chemistry at Cambridge University in Britain before joining Cipla, which was founded by his father.
“My father never forced me but chemistry was my best subject,” said Hamied, who became chairman of the company in 1989.
His bold step in offering cheap AIDS drugs turned out to be a smart business move.
Cipla is now the world’s largest AIDS antiretroviral drugs supplier and the publicly listed company is valued at nearly US$5 billion, while business magazine Forbes puts Hamied’s personal fortune at US$1.75 billion.
However, Hamied said poverty-racked India “can’t afford to divide people into those who can afford life-saving drugs and those who can’t.”
He believes the pharma giants should let emerging market drugmakers make copycat medicines in exchange for small royalties.
Some 95 percent of Western firms’ profits come from regulated developed markets like Japan, Europe and the US, so the pharmaceutical giants “really won’t lose out,” he added.
Even with the reduced price of generic drugs, such medicines are still beyond the reach of many of the world’s poorest, said Hamied, who confesses he has his eye on his legacy.
“I want it to be said when I leave this world that ‘he was not just a money-making machine,’” he said.

SEEKING CHANGE: A hospital worker said she did not vote in previous elections, but ‘now I can see that maybe my vote can change the system and the country’ Voting closed yesterday across the Solomon Islands in the south Pacific nation’s first general election since the government switched diplomatic allegiance from Taiwan to Beijing and struck a secret security pact that has raised fears of the Chinese navy gaining a foothold in the region. The Solomon Islands’ closer relationship with China and a troubled domestic economy weighed on voters’ minds as they cast their ballots. As many as 420,000 registered voters had their say across 50 national seats. For the first time, the national vote also coincided with elections for eight of the 10 local governments. Esther Maeluma cast her vote in the
Nearly half of China’s major cities are suffering “moderate to severe” levels of subsidence, putting millions of people at risk of flooding, especially as sea levels rise, according to a study of nationwide satellite data released yesterday. The authors of the paper, published by the journal Science, found that 45 percent of China’s urban land was sinking faster than 3mm per year, with 16 percent at more than 10mm per year, driven not only by declining water tables, but also the sheer weight of the built environment. With China’s urban population already in excess of 900 million people, “even a small portion
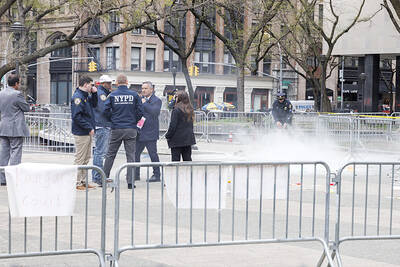
UNSETTLING IMAGES: The scene took place in front of TV crews covering the Trump trial, with a CNN anchor calling it an ‘emotional and unbelievably disturbing moment’ A man who doused himself in an accelerant and set himself on fire outside the courthouse where former US president Donald Trump is on trial has died, police said yesterday. The New York City Police Department (NYPD) said the man was declared dead by staff at an area hospital. The man was in Collect Pond Park at about 1:30pm on Friday when he took out pamphlets espousing conspiracy theories, tossed them around, then doused himself in an accelerant and set himself on fire, officials and witnesses said. A large number of police officers were nearby when it happened. Some officers and bystanders rushed

HYPOCRISY? The Chinese Ministry of Foreign Affairs yesterday asked whether Biden was talking about China or the US when he used the word ‘xenophobic’ US President Joe Biden on Wednesday called for a hike in steel tariffs on China, accusing Beijing of cheating as he spoke at a campaign event in Pennsylvania. Biden accused China of xenophobia, too, in a speech to union members in Pittsburgh. “They’re not competing, they’re cheating. They’re cheating and we’ve seen the damage here in America,” Biden said. Chinese steel companies “don’t need to worry about making a profit because the Chinese government is subsidizing them so heavily,” he said. Biden said he had called for the US Trade Representative to triple the tariff rates for Chinese steel and aluminum if Beijing was