Scientists have established the first case of the new H1N1 influenza strain showing resistance to Tamiflu, the main antiviral flu drug, Danish officials and the manufacturer said on Monday.
“While receiving the drug, the patient appeared to develop resistance to it,” David Reddy, Roche Holding AG’s pandemic taskforce leader, told reporters on a conference call on a case observed in Denmark. “This is the first report we have of it in H1N1.”
Common seasonal flu can resist Tamiflu and Reddy said a case of resistance in influenza A(H1N1) — also know as swine flu — was not unexpected. Roche had been working on strategies to counter such a development.
The WHO declared an influenza pandemic earlier this month and advised governments to prepare for a long-term battle against an unstoppable new flu virus. The WHO had no immediate comment on the case of Tamiflu resistance.
The UN agency raised its pandemic flu alert to phase 6 on a six-point scale, indicating the first influenza pandemic since 1968 is under way.
“The person is well now and no further contagion with the resistant virus has been detected,” Denmark’s State Serum Institute said in a statement.
Roche shares closed up 1.4 percent at 149 Swiss francs on Monday, outperforming a 0.7 percent firmer DJ STOXX European drugs index.
This case does not change the recommendation to use Tamiflu, the institute said.
The US Centers for Disease Control and Prevention has expressed concern the new strain may mix with seasonal flu strains, which are still circulating.
The WHO has said Tamiflu was working against strains of the new H1N1 flu, but some analysts have expressed concern it might be less effective than Relenza, GlaxoSmithKline’s inhaled drug, since there have been widespread reports of resistance by seasonal H1N1 flu.
The seasonal strain of H1N1 is a distant cousin of the swine flu and was widely resistant to Tamiflu this year.

‘IN A DIFFERENT PLACE’: The envoy first visited Shanghai, where he attended a Chinese basketball playoff match, and is to meet top officials in Beijing tomorrow US Secretary of State Antony Blinken yesterday arrived in China on his second visit in a year as the US ramps up pressure on its rival over its support for Russia while also seeking to manage tensions with Beijing. The US diplomat tomorrow is to meet China’s top brass in Beijing, where he is also expected to plead for restraint as Taiwan inaugurates president-elect William Lai (賴清德), and to raise US concerns on Chinese trade practices. However, Blinken is also seeking to stabilize ties, with tensions between the world’s two largest economies easing since his previous visit in June last year. At the
Nearly half of China’s major cities are suffering “moderate to severe” levels of subsidence, putting millions of people at risk of flooding, especially as sea levels rise, according to a study of nationwide satellite data released yesterday. The authors of the paper, published by the journal Science, found that 45 percent of China’s urban land was sinking faster than 3mm per year, with 16 percent at more than 10mm per year, driven not only by declining water tables, but also the sheer weight of the built environment. With China’s urban population already in excess of 900 million people, “even a small portion
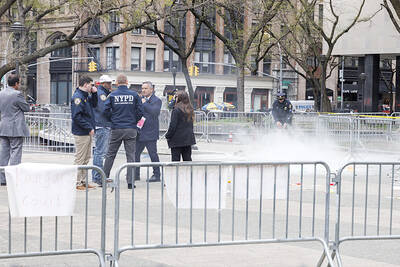
UNSETTLING IMAGES: The scene took place in front of TV crews covering the Trump trial, with a CNN anchor calling it an ‘emotional and unbelievably disturbing moment’ A man who doused himself in an accelerant and set himself on fire outside the courthouse where former US president Donald Trump is on trial has died, police said yesterday. The New York City Police Department (NYPD) said the man was declared dead by staff at an area hospital. The man was in Collect Pond Park at about 1:30pm on Friday when he took out pamphlets espousing conspiracy theories, tossed them around, then doused himself in an accelerant and set himself on fire, officials and witnesses said. A large number of police officers were nearby when it happened. Some officers and bystanders rushed

Beijing is continuing to commit genocide and crimes against humanity against Uyghurs and other Muslim minorities in its western Xinjiang province, U.S. Secretary of State Antony Blinken said in a report published on Monday, ahead of his planned visit to China this week. The State Department’s annual human rights report, which documents abuses recorded all over the world during the previous calendar year, repeated language from previous years on the treatment of Muslims in Xinjiang, but the publication raises the issue ahead of delicate talks, including on the war in Ukraine and global trade, between the top U.S. diplomat and Chinese