Health experts and drugs companies on Friday urged rich countries to come up with public funding to spur development of a human vaccine against a pandemic strain of the bird flu virus that could kill millions of people around the world within months.
"We would hope that one of the major messages from this meeting to governments, to support vaccine testing, is going to get across," the World Health Organization's senior flu specialist Klaus Stoehr told journalists after a meeting at the WHO.
Stoehr said that the gathering of 50 top executives from pharmaceutical companies, public health regulators and government officials had agreed to boost cooperation on the issue.
But he warned that private industry alone could not invest about 11 million euros needed in the short term to develop the candidate vaccine needed to inoculate six to seven billion people worldwide in an emergency.
"Without money nothing is going to move with the pandemic vaccine," Stoehr said, emphasizing that 90 percent of the production capacity was in industrialized countries.
Fears of the worldwide spread of a more virulent and deadly strain of influenza have been heightened by recent cases of the recently discovered H5N1 strain of avian flu in Thailand and Vietnam, where the disease has killed 32 people this year.
The WHO estimated that it would take three to six months for a mutated virus to travel around the world, with 25 to 30 percent of the world's population likely to be infected.
About one percent of those who fell ill were likely to die, Stoehr said.
That compared to 2.6 percent death rate during the 1918 flu pandemic, which killed an estimated 21 million people when the world's population was far smaller and international travel was less widespread.
"Practically everybody" -- 6.2 billion people -- would need to be vaccinated in the event of a pandemic, Stoehr added.
Flu vaccines were not available in the three pandemics that hit the world in the last century, most recently in 1968.
Current production capacity for a flu vaccine was about 300 million doses, reaching maybe up to 1.2 billion in a year in an emergency, he estimated.
If development was stretched beyond the two companies - Aventis-Pasteur and Chiron -- currently developing a candidate vaccine for a pandemic strain, it would be possible to produce six billion doses in a year in the best case, the experts estimated.
"Market forces have not brought companies into pandemic vaccine development," Stoehr noted.
Boosted development was also necessary to ensure that the 90 percent of the production capacity located in countries with 12 percent of the world's population would also serve other areas in an emergency, the WHO said.
Luc Hassel of pharmaceutical giant Aventis-Pasteur, and head of an industry task force on regular flu vaccines, said public funding was needed to overcome the lack of a commercial return on pandemic research.
"One of the key messages that we heard in the past two days is that there should be shared responsibilities, otherwise companies will have limited capacity to develop extensive research in this field," he said.
Only the United States has so far invested in pandemic vaccine development.
The Japanese government revealed that it was prepared to support tests on another candidate vaccine by local companies, while France was exploring ways of securing a national pandemic vaccine stockpile.
The meeting agreed to set up a working group involving the WHO and the US Food and Drug Administration to find ways to coordinate vaccine approval by regulators, potentially knocking months off development times.
"There are pathways... but there are a few glitches," said Arlene King of Canada's public health authority.
Nearly half of China’s major cities are suffering “moderate to severe” levels of subsidence, putting millions of people at risk of flooding, especially as sea levels rise, according to a study of nationwide satellite data released yesterday. The authors of the paper, published by the journal Science, found that 45 percent of China’s urban land was sinking faster than 3mm per year, with 16 percent at more than 10mm per year, driven not only by declining water tables, but also the sheer weight of the built environment. With China’s urban population already in excess of 900 million people, “even a small portion
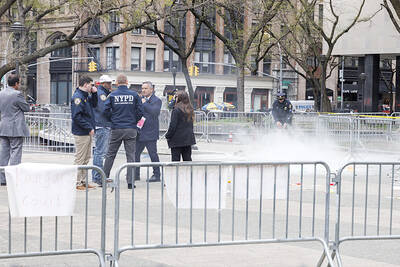
UNSETTLING IMAGES: The scene took place in front of TV crews covering the Trump trial, with a CNN anchor calling it an ‘emotional and unbelievably disturbing moment’ A man who doused himself in an accelerant and set himself on fire outside the courthouse where former US president Donald Trump is on trial has died, police said yesterday. The New York City Police Department (NYPD) said the man was declared dead by staff at an area hospital. The man was in Collect Pond Park at about 1:30pm on Friday when he took out pamphlets espousing conspiracy theories, tossed them around, then doused himself in an accelerant and set himself on fire, officials and witnesses said. A large number of police officers were nearby when it happened. Some officers and bystanders rushed

Beijing is continuing to commit genocide and crimes against humanity against Uyghurs and other Muslim minorities in its western Xinjiang province, U.S. Secretary of State Antony Blinken said in a report published on Monday, ahead of his planned visit to China this week. The State Department’s annual human rights report, which documents abuses recorded all over the world during the previous calendar year, repeated language from previous years on the treatment of Muslims in Xinjiang, but the publication raises the issue ahead of delicate talks, including on the war in Ukraine and global trade, between the top U.S. diplomat and Chinese

HYPOCRISY? The Chinese Ministry of Foreign Affairs yesterday asked whether Biden was talking about China or the US when he used the word ‘xenophobic’ US President Joe Biden on Wednesday called for a hike in steel tariffs on China, accusing Beijing of cheating as he spoke at a campaign event in Pennsylvania. Biden accused China of xenophobia, too, in a speech to union members in Pittsburgh. “They’re not competing, they’re cheating. They’re cheating and we’ve seen the damage here in America,” Biden said. Chinese steel companies “don’t need to worry about making a profit because the Chinese government is subsidizing them so heavily,” he said. Biden said he had called for the US Trade Representative to triple the tariff rates for Chinese steel and aluminum if Beijing was