In the wake of recent food scares, lawmakers yesterday again proposed the establishment of a food safety police force and called for reinstating the registration system for compound food additives abolished in 2000.
Food safety and sanitation inspectors, unlike police and prosecutors, do not have the power to search factories or seize products.
Chang Chi Foodstuff Factory Co, the company at the center of the adulterated oil scandal, obstructed inspections by local health authorities several times before the Ministry of Justice’s prosecution and investigation unit intervened, the Ministry of Health and Welfare said.

Photo: Lin Cheng-kung, Taipei Times
Democratic Progressive Party Legislator Tien Chiu-chin (田秋堇) slammed the health ministry yesterday in the legislature’s plenary session for failing to come up with a plan to establish a food police force, as previously recommend by legislative committees.
She also asked the health ministry to present an evaluation report on the proposal within two weeks.
Minister of Health and Welfare Chiu Wen-ta (邱文達) responded to Tien’s request by saying the health ministry, together with the National Police Agency, has been working on the idea, but efforts have been hampered by manpower shortages.
Chiu and Chinese Nationalist Party (KMT) Legislator Tsai Chin-lung (蔡錦隆) also faulted the health ministry for letting food companies off the hook by no longer requiring them to register compound food additives and flavorings, which they said created a “food safety loophole” that contributed to violations.
Food and Drug Administration’s (FDA) Food Division director Tsai Shu-chen (蔡淑貞) said the registration system was scrapped to streamline trade with foreign countries after Taiwan joined the WTO in 2000.
She said that the health ministry has given notice of a new regulation that will require all food-related companies to register their products.
However, Tien said this only require firms to list product names, meaning consumers and downstream companies will still be exposed.
“Whether consumers and downstream businesses suffer depends on how conscientious upstream food companies are. Yet [Chiu] also said earlier [that the system] cannot beat human evil. Do you still think the current system is sufficient?” Tien asked FDA Director-General Yeh Ming-kung (葉明功).
Yeh said the agency would seriously consider the proposal to restore the registration system for compound food additives.
In related developments, several major edible oil distributors in China’s Fujian Province said they will file a lawsuit against Chang Chi, a lawyer said yesterday.
Tsai Wen-pin (蔡文斌), who is representing the companies in Xiamen, China, said he would file the suit today and seek damages of at least NT$5.3 million (US$179,600). Of that, NT$1.9 million would be for inventory losses, while 700,000 yuan (US$115,000) is for the damage done to his clients’ reputations.
The lawyer said the amount of compensation requested may go up, because the distributors have not given him a final figure on losses incurred yet as Chang Chi products are still being returned by customers. The final claim would be made after spoken proceedings, he said.
Tsai Wen-pin previously said that cross-strait lawsuits are rarely filed as it is difficult to be awarded compensation in such cases.
“Realistically speaking, the assets of the manufacturer in question have been frozen, so even if we win the case, it would be difficult for us to obtain compensation,” Tsai said.
Last month, Chang Chi was found to have been mixing cottonseed oil and other cheap oils into its more expensive grapeseed and olive oil products. In some cases, it used copper chlorophyllin, a coloring agent, to make the substitutes look more like olive oil.
Other oil suppliers subsequently found to be selling similarly adulterated oil include Flavor Full Foods Inc, Formosa Oilseed Processing Co and Wei Chuan Foods Corp.

THE HAWAII FACTOR: While a 1965 opinion said an attack on Hawaii would not trigger Article 5, the text of the treaty suggests the state is covered, the report says NATO could be drawn into a conflict in the Taiwan Strait if Chinese forces attacked the US mainland or Hawaii, a NATO Defense College report published on Monday says. The report, written by James Lee, an assistant research fellow at Academia Sinica’s Institute of European and American Studies, states that under certain conditions a Taiwan contingency could trigger Article 5 of NATO, under which an attack against any member of the alliance is considered an attack against all members, necessitating a response. Article 6 of the North Atlantic Treaty specifies that an armed attack in the territory of any member in Europe,
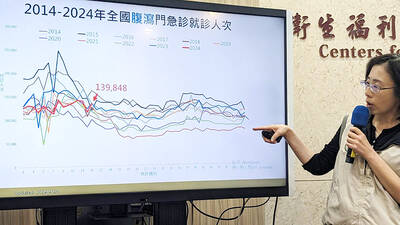
FLU SEASON: Twenty-six severe cases were reported from Tuesday last week to Monday, including a seven-year-old girl diagnosed with influenza-associated encephalopathy Nearly 140,000 people sought medical assistance for diarrhea last week, the Centers for Disease Control (CDC) said on Tuesday. From April 7 to Saturday last week, 139,848 people sought medical help for diarrhea-related illness, a 15.7 percent increase from last week’s 120,868 reports, CDC Epidemic Intelligence Center Deputy Director Lee Chia-lin (李佳琳) said. The number of people who reported diarrhea-related illness last week was the fourth highest in the same time period over the past decade, Lee said. Over the past four weeks, 203 mass illness cases had been reported, nearly four times higher than the 54 cases documented in the same period
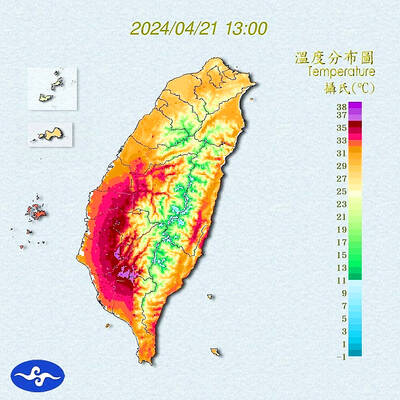
Heat advisories were in effect for nine administrative regions yesterday afternoon as warm southwesterly winds pushed temperatures above 38°C in parts of southern Taiwan, the Central Weather Administration (CWA) said. As of 3:30pm yesterday, Tainan’s Yujing District (玉井) had recorded the day’s highest temperature of 39.7°C, though the measurement will not be included in Taiwan’s official heat records since Yujing is an automatic rather than manually operated weather station, the CWA said. Highs recorded in other areas were 38.7°C in Kaohsiung’s Neimen District (內門), 38.2°C in Chiayi City and 38.1°C in Pingtung’s Sandimen Township (三地門), CWA data showed. The spell of scorching

HOSPITALITY HIT: Hotels in Hualien have an occupancy rate of 10 percent, down from 30 percent before the earthquake, a Tourism Administration official said The Executive Yuan yesterday unveiled a stimulus package of vouchers and subsidies to revive tourism in Hualien County following a quake measuring 7.2 on the Richter scale. The tremor on April 3, which killed at least 17 people and left two others missing, caused the county an estimated NT$3 billion (US$92.7 million) in damages. The Ministry of Economic Affairs is to issue vouchers worth NT$200 at the price of NT$100 for purchases at the Dongdamen Night Market (東大門夜市) in Hualien City to boost spending, a ministry official told a news conference after a Cabinet meeting in Taipei. The ministry plans to issue 18,400