The Executive Yuan yesterday approved draft legislation regulating the establishment of human biological databases that would require providers of genetic information to be informed of how the data might be used.
Cabinet spokesman Su Jun-pin (蘇俊賓) told a press conference after the Executive Yuan’s weekly meeting that the Department of Health (DOH) proposed the bill as part of a response to ethical issues.
He said it constituted a significant step in protecting the privacy of those who provide genetic samples for medical research.
The bill will be submitted to the Legislative Yuan for review.
COMMITTEE
If the legislature passes the bill, it would authorize the DOH to issue permits to entities that file applications for establishing human biological databanks and require that the founders of the databases to set up a committee in charge of ethical issues.
Those who fail to obtain a permit would be fined between NT$1 million (US$30,000) and NT$5 million, while the genetic samples in the databanks would be destroyed.
Under the bill, only adults would be allowed to give biological samples, with the exception of members of specific groups, such as patients suffering from rare genetic disorders.
Participants in the genetic pools would have the right to know how their samples would be used, how long they would remain in research labs, possible complications that may take place during collection of the samples or the risks to the providers and their family members as a result of information gleaned from DNA data.
CONTRACTS
Biological databanks would be allowed to store and use the genetic information of a deceased participant unless existing contracts state otherwise.
The draft would also empower participants to withdraw their samples from databases.
All personnel involved in the databases would have to maintain confidentiality, the draft said.
Those who use the genetic information for purposes other than medical research would be liable to fines of NT$300,000 to NT$1.5 million.
Biological databases established prior to the legislature’s passage of the bill will be required to file applications at the DOH and follow due procedures stated in the draft within one year.
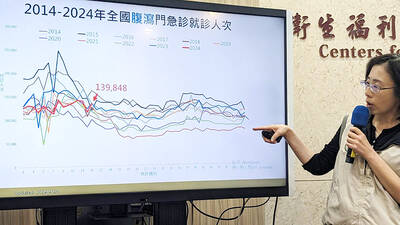
FLU SEASON: Twenty-six severe cases were reported from Tuesday last week to Monday, including a seven-year-old girl diagnosed with influenza-associated encephalopathy Nearly 140,000 people sought medical assistance for diarrhea last week, the Centers for Disease Control (CDC) said on Tuesday. From April 7 to Saturday last week, 139,848 people sought medical help for diarrhea-related illness, a 15.7 percent increase from last week’s 120,868 reports, CDC Epidemic Intelligence Center Deputy Director Lee Chia-lin (李佳琳) said. The number of people who reported diarrhea-related illness last week was the fourth highest in the same time period over the past decade, Lee said. Over the past four weeks, 203 mass illness cases had been reported, nearly four times higher than the 54 cases documented in the same period

A group of Taiwanese-American and Tibetan-American students at Harvard University on Saturday disrupted Chinese Ambassador to the US Xie Feng’s (謝鋒) speech at the school, accusing him of being responsible for numerous human rights violations. Four students — two Taiwanese Americans and two from Tibet — held up banners inside a conference hall where Xie was delivering a speech at the opening ceremony of the Harvard Kennedy School China Conference 2024. In a video clip provided by the Coalition of Students Resisting the CCP (Chinese Communist Party), Taiwanese-American Cosette Wu (吳亭樺) and Tibetan-American Tsering Yangchen are seen holding banners that together read:

UNAWARE: Many people sit for long hours every day and eat unhealthy foods, putting them at greater risk of developing one of the ‘three highs,’ an expert said More than 30 percent of adults aged 40 or older who underwent a government-funded health exam were unaware they had at least one of the “three highs” — high blood pressure, high blood lipids or high blood sugar, the Health Promotion Administration (HPA) said yesterday. Among adults aged 40 or older who said they did not have any of the “three highs” before taking the health exam, more than 30 percent were found to have at least one of them, Adult Preventive Health Examination Service data from 2022 showed. People with long-term medical conditions such as hypertension or diabetes usually do not
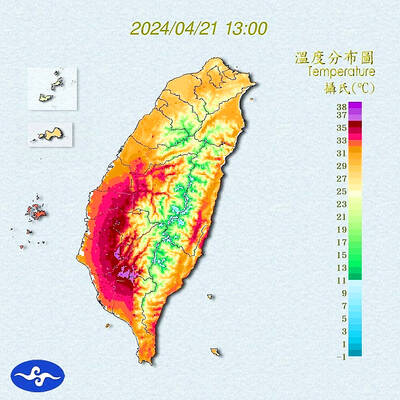
Heat advisories were in effect for nine administrative regions yesterday afternoon as warm southwesterly winds pushed temperatures above 38°C in parts of southern Taiwan, the Central Weather Administration (CWA) said. As of 3:30pm yesterday, Tainan’s Yujing District (玉井) had recorded the day’s highest temperature of 39.7°C, though the measurement will not be included in Taiwan’s official heat records since Yujing is an automatic rather than manually operated weather station, the CWA said. Highs recorded in other areas were 38.7°C in Kaohsiung’s Neimen District (內門), 38.2°C in Chiayi City and 38.1°C in Pingtung’s Sandimen Township (三地門), CWA data showed. The spell of scorching