A team of scientists from the National Science Council have found that a human growth factor that triggers the release of stem cells from bone marrow shows potential as a treatment for Alzheimer's disease and other degenerative brain disorders.
Shen Che-kun (
This is the first piece of research that applies G-CSF therapy, most often used to accelerate recovery from chemotherapy, to Alzheimer's disease, he said.
"Studies conducted on mice show that G-CSF not only arrests deterioration in mental capacity but allows the affected mice to recover lost mental capacity to levels comparable to normal mice of the same age," Shen said.
The team used two AD mouse models with Alzheimer's dislike symptoms -- one was generated by injecting the brains of normal mice with beta-amyloid protein and another was a strain of transgenic mice which naturally exhibit Alzheimer's disease-like neuronal apoptosis and memory loss.
The study, co-authored by Shen and Tsai Kuen-jer (
G-CSF is a growth factor that is naturally present in the body in small quantities and is thought to be linked with the regeneration of brain tissue, Shen said.
During their study, researchers injected G-CSF directly into the mice's thorax. Its presence in the bloodstream facilitated the release of hematopoietic stem cells from bone marrows, Shen said.
Although Shen and Tsai said that they have not conclusively proved the mechanism by which the damage was repaired, Shen said that he thought it was most likely that the stem cells released by the G-CSF injection from the bone marrow passed into the brain from the bloodstream, where they attached to sites of damage and became differentiated into new cells.
"There have been previous studies where injections of G-CSF into the bloodstream was found to repair damaged heart tissue," Shen said. "In our case, we saw that the G-CSF injection appeared to cause new cells to grow where the neuron damage was the greatest."
If successful, G-CSF therapy could open up a new avenue of Alzheimer's treatment that is less invasive and more effective than current therapies, Shen said.
"However, remember that it has only been found effective in mice so far," cautioned Shen, who said that further trials on human subjects were needed to determine the treatment's efficacy and safety.
If the drug proves to be effective in clinical testing, it may become available on the market in as little as five years, the researchers said.
Because G-CSF is already widely used to treat neutropenia in chemotherapy patients, the drug does not have to undergo pre-clinical toxicological testing or phase I clinical testing.

A group of Taiwanese-American and Tibetan-American students at Harvard University on Saturday disrupted Chinese Ambassador to the US Xie Feng’s (謝鋒) speech at the school, accusing him of being responsible for numerous human rights violations. Four students — two Taiwanese Americans and two from Tibet — held up banners inside a conference hall where Xie was delivering a speech at the opening ceremony of the Harvard Kennedy School China Conference 2024. In a video clip provided by the Coalition of Students Resisting the CCP (Chinese Communist Party), Taiwanese-American Cosette Wu (吳亭樺) and Tibetan-American Tsering Yangchen are seen holding banners that together read:

UNAWARE: Many people sit for long hours every day and eat unhealthy foods, putting them at greater risk of developing one of the ‘three highs,’ an expert said More than 30 percent of adults aged 40 or older who underwent a government-funded health exam were unaware they had at least one of the “three highs” — high blood pressure, high blood lipids or high blood sugar, the Health Promotion Administration (HPA) said yesterday. Among adults aged 40 or older who said they did not have any of the “three highs” before taking the health exam, more than 30 percent were found to have at least one of them, Adult Preventive Health Examination Service data from 2022 showed. People with long-term medical conditions such as hypertension or diabetes usually do not
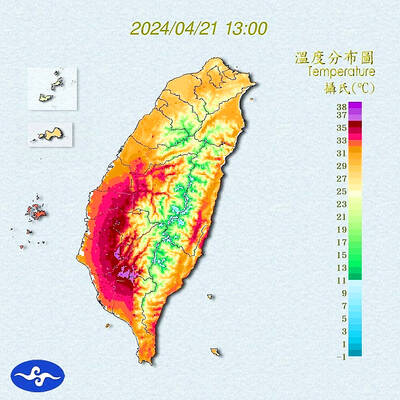
Heat advisories were in effect for nine administrative regions yesterday afternoon as warm southwesterly winds pushed temperatures above 38°C in parts of southern Taiwan, the Central Weather Administration (CWA) said. As of 3:30pm yesterday, Tainan’s Yujing District (玉井) had recorded the day’s highest temperature of 39.7°C, though the measurement will not be included in Taiwan’s official heat records since Yujing is an automatic rather than manually operated weather station, the CWA said. Highs recorded in other areas were 38.7°C in Kaohsiung’s Neimen District (內門), 38.2°C in Chiayi City and 38.1°C in Pingtung’s Sandimen Township (三地門), CWA data showed. The spell of scorching

POLICE INVESTIGATING: A man said he quit his job as a nurse at Taipei Tzu Chi Hospital as he had been ‘disgusted’ by the behavior of his colleagues A man yesterday morning wrote online that he had witnessed nurses taking photographs and touching anesthetized patients inappropriately in Taipei Tzu Chi Hospital’s operating theaters. The man surnamed Huang (黃) wrote on the Professional Technology Temple bulletin board that during his six-month stint as a nurse at the hospital, he had seen nurses taking pictures of patients, including of their private parts, after they were anesthetized. Some nurses had also touched patients inappropriately and children were among those photographed, he said. Huang said this “disgusted” him “so much” that “he felt the need to reveal these unethical acts in the operating theater