HANDSETS
Silitech to reduce capital
Handset keypad supplier Silitech Technology Corp’s (閎暉) board on Monday approved a proposal to reduce the company’s capital by 66.55 percent to refund cash to shareholders and adjust the company’s capital structure to increase returns on equity. In a regulatory filing, Silitech said it would cut its capital by NT$1.19 billion (US$38.7 million) by canceling 119.38 million shares and return NT$6.66 per share to shareholders. The company also decided not to propose distributing a cash dividend for last year because of losses, although net losses per share narrowed from NT$0.46 in 2017 to NT$0.19 last year. Revenue decreased 1.49 percent year-on-year to NT$2.25 billion.
COMPUTERS
Getac to pay NT$3 dividend
Rugged PC vendor Getac Technology Corp (神基) said its board of directors yesterday approved a proposal to distribute a cash dividend of NT$3 per share. The dividend payout was based on the company’s earnings of NT$2.21 billion last year, or earnings per share of NT$3.83, the company said in a filing with the Taiwan Stock Exchange. The proposed cash dividend translates to a payout ratio of 78.33 percent and a dividend yield of 6.41 percent based on stock’s closing price of NT$46.8 yesterday. The dividend proposal is to be voted on at the annual shareholders’ meeting on May 31.
PHARMACEUTICALS
SynCore enrolls US patient
SynCore Biotechnology Co (杏國新藥) yesterday announced that SB05PC, its new pancreatic cancer drug, has enrolled its first patient in the US for a global phase III clinical trial across seven nations. The company said that progress made with the US Food and Drug Administration would help with its approval efforts in other markets and that SB05PC has been granted orphan drug status due to a lack of available alternative second-line treatments. Last month, the company enrolled patients in Hungary, Israel and France. It said that its patient enrollment is on track and expected to produce an interim report in the second half of this year.
PHARMACEUTICALS
Milestone payment received
Anxo Pharmaceutical Co Ltd (瑩碩生技) has received its first milestone payment from its unidentified Chinese partner in Guangzhou, the company said yesterday. Anxo did not reveal the amount of the milestone payment. The company has inked collaborations with six Chinese companies to work on 18 new drug projects and they are aiming to take advantage of ongoing regulatory reforms in China to expand the availability of generic drugs in the nation. Drug developers are heading toward a cutthroat battle to make it into the top three for a certain drug class as China adopts harmonized approval standards and requirements. Anxo said that it and the Guangzhou-based company are working on two cardiovascular drugs and a pediatric respiratory drug with a combined potential market value of about 10.5 billion yuan (US$1.57 billion).
MONEY SUPPLY
M1B grows 6.55 percent
Last month’s M1B — a measure of money in circulation — grew 6.55 percent year-on-year, accelerating from a 5.69 percent increase the previous month, while M2 — which includes the M1B, time deposits, foreign-currency deposits and mutual funds — increased 3.14 percent from 3.07 percent the previous month, mainly because of growth in bank loans, investments and net foreign capital inflows, the central bank said in a statement yesterday.

Stephen Garrett, a 27-year-old graduate student, always thought he would study in China, but first the country’s restrictive COVID-19 policies made it nearly impossible and now he has other concerns. The cost is one deterrent, but Garrett is more worried about restrictions on academic freedom and the personal risk of being stranded in China. He is not alone. Only about 700 American students are studying at Chinese universities, down from a peak of nearly 25,000 a decade ago, while there are nearly 300,000 Chinese students at US schools. Some young Americans are discouraged from investing their time in China by what they see
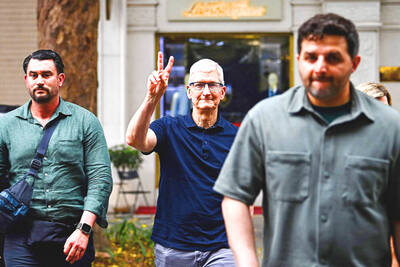
MAJOR DROP: CEO Tim Cook, who is visiting Hanoi, pledged the firm was committed to Vietnam after its smartphone shipments declined 9.6% annually in the first quarter Apple Inc yesterday said it would increase spending on suppliers in Vietnam, a key production hub, as CEO Tim Cook arrived in the country for a two-day visit. The iPhone maker announced the news in a statement on its Web site, but gave no details of how much it would spend or where the money would go. Cook is expected to meet programmers, content creators and students during his visit, online newspaper VnExpress reported. The visit comes as US President Joe Biden’s administration seeks to ramp up Vietnam’s role in the global tech supply chain to reduce the US’ dependence on China. Images on

Taiwan Transport and Storage Corp (TTS, 台灣通運倉儲) yesterday unveiled its first electric tractor unit — manufactured by Volvo Trucks — in a ceremony in Taipei, and said the unit would soon be used to transport cement produced by Taiwan Cement Corp (TCC, 台灣水泥). Both TTS and TCC belong to TCC International Holdings Ltd (台泥國際集團). With the electric tractor unit, the Taipei-based cement firm would become the first in Taiwan to use electric vehicles to transport construction materials. TTS chairman Koo Kung-yi (辜公怡), Volvo Trucks vice president of sales and marketing Johan Selven, TCC president Roman Cheng (程耀輝) and Taikoo Motors Group

New apartments in Taiwan’s major cities are getting smaller, while old apartments are increasingly occupied by older people, many of whom live alone, government data showed. The phenomenon has to do with sharpening unaffordable property prices and an aging population, property brokers said. Apartments with one bedroom that are two years old or older have gained a noticeable presence in the nation’s six special municipalities as well as Hsinchu county and city in the past five years, Evertrust Rehouse Co (永慶房產集團) found, citing data from the government’s real-price transaction platform. In Taipei, apartments with one bedroom accounted for 19 percent of deals last