If the UK cannot reach an accord regarding new drug approvals by the European Medicines Agency (EMA) following Brexit, the government might consider looking to the US for support.
“That is not our desired outcome,” British lawmaker James O’Shaughnessy said at the Bio-Europe conference in Berlin on Monday.
The UK wants to continue to benefit from its relationship with the EMA, he said, but added that “there are approvals being made by other stringent regulators.”
At issue is the question of whether the UK can avoid the expense and challenges involved in taking over the task of reviewing and approving hundreds of drugs for use in the country once it leaves the EU in March 2019.
Pharmaceutical industry executives have advocated reaching a pact that would essentially allow for mutual recognition of new medicines between the EMA and the British Medicines and Healthcare products Regulatory Agency, thereby eliminating the need to replicate processes and reducing costs for drugmakers.
Without such a deal, companies might opt to introduce new drugs in the UK, home to about 66 million people, only after they have tapped the US, continental Europe and other, bigger markets.
The EMA is poised to move away from its London headquarters and end its oversight of the British market after Brexit.
The US Food and Drug Administration (FDA) is sometimes faster than the EMA in approving new medicines, and FDA Commissioner Scott Gottlieb aims to speed up the process even further.

Stephen Garrett, a 27-year-old graduate student, always thought he would study in China, but first the country’s restrictive COVID-19 policies made it nearly impossible and now he has other concerns. The cost is one deterrent, but Garrett is more worried about restrictions on academic freedom and the personal risk of being stranded in China. He is not alone. Only about 700 American students are studying at Chinese universities, down from a peak of nearly 25,000 a decade ago, while there are nearly 300,000 Chinese students at US schools. Some young Americans are discouraged from investing their time in China by what they see
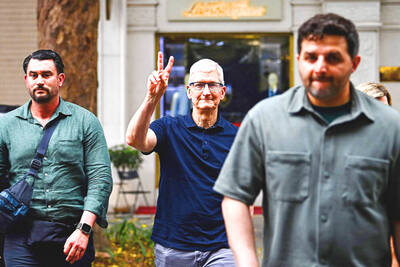
MAJOR DROP: CEO Tim Cook, who is visiting Hanoi, pledged the firm was committed to Vietnam after its smartphone shipments declined 9.6% annually in the first quarter Apple Inc yesterday said it would increase spending on suppliers in Vietnam, a key production hub, as CEO Tim Cook arrived in the country for a two-day visit. The iPhone maker announced the news in a statement on its Web site, but gave no details of how much it would spend or where the money would go. Cook is expected to meet programmers, content creators and students during his visit, online newspaper VnExpress reported. The visit comes as US President Joe Biden’s administration seeks to ramp up Vietnam’s role in the global tech supply chain to reduce the US’ dependence on China. Images on

Taiwan Transport and Storage Corp (TTS, 台灣通運倉儲) yesterday unveiled its first electric tractor unit — manufactured by Volvo Trucks — in a ceremony in Taipei, and said the unit would soon be used to transport cement produced by Taiwan Cement Corp (TCC, 台灣水泥). Both TTS and TCC belong to TCC International Holdings Ltd (台泥國際集團). With the electric tractor unit, the Taipei-based cement firm would become the first in Taiwan to use electric vehicles to transport construction materials. TTS chairman Koo Kung-yi (辜公怡), Volvo Trucks vice president of sales and marketing Johan Selven, TCC president Roman Cheng (程耀輝) and Taikoo Motors Group

New apartments in Taiwan’s major cities are getting smaller, while old apartments are increasingly occupied by older people, many of whom live alone, government data showed. The phenomenon has to do with sharpening unaffordable property prices and an aging population, property brokers said. Apartments with one bedroom that are two years old or older have gained a noticeable presence in the nation’s six special municipalities as well as Hsinchu county and city in the past five years, Evertrust Rehouse Co (永慶房產集團) found, citing data from the government’s real-price transaction platform. In Taipei, apartments with one bedroom accounted for 19 percent of deals last