Lotus Pharmaceuticals Co (美時化學製藥) on Tuesday said it has successfully challenged a patent claim that has been blocking the company’s efforts to commercialize a generic drug.
The US District Court for the District of Delaware ruled that a final remaining patent covering Uceris, a US brand name for an ulcerative colitis drug based on budesonide, was not infringed upon by Lotus, the company said.
The court challenge was overseen by US-based drugmaker Alvogen Group Inc, Lotus said in a statement.
Alvogen and Lotus are to file an abbreviated new drug application with the US Food and Drug Administration for a generic version of Uceris, while Alvogen is prepared to contest appeals by Valeant Pharmaceuticals International Inc, owner of the drug’s original developer.
The dispute began in February 2015, when Alvogen was sued by Salix Pharmaceuticals Inc, the original developer of Uceris, for filing a paragraph IV challenge to certify that patents protecting the drug are invalid and unenforceable.
The dispute continued after Salix was in April 2015 acquired by Valeant, which took over the rights to market Uceris.
The agency could grant Alvogen and Lotus approval at the end of a 180-day exclusivity period granted to Teva Pharmaceutical Industries Ltd, which was the first company to file a generic variant of the drug, Lotus said.
Over the past 12 months, Uceris, which as yet has no generic competitors in the US, commanded a market valued at about US$200 million in that country, Lotus said, citing findings by healthcare researcher IMS Health Inc.

Stephen Garrett, a 27-year-old graduate student, always thought he would study in China, but first the country’s restrictive COVID-19 policies made it nearly impossible and now he has other concerns. The cost is one deterrent, but Garrett is more worried about restrictions on academic freedom and the personal risk of being stranded in China. He is not alone. Only about 700 American students are studying at Chinese universities, down from a peak of nearly 25,000 a decade ago, while there are nearly 300,000 Chinese students at US schools. Some young Americans are discouraged from investing their time in China by what they see
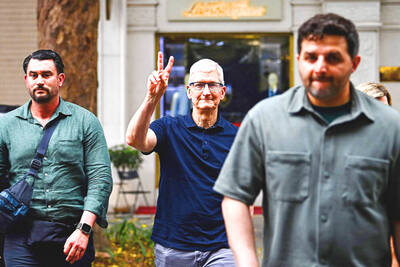
MAJOR DROP: CEO Tim Cook, who is visiting Hanoi, pledged the firm was committed to Vietnam after its smartphone shipments declined 9.6% annually in the first quarter Apple Inc yesterday said it would increase spending on suppliers in Vietnam, a key production hub, as CEO Tim Cook arrived in the country for a two-day visit. The iPhone maker announced the news in a statement on its Web site, but gave no details of how much it would spend or where the money would go. Cook is expected to meet programmers, content creators and students during his visit, online newspaper VnExpress reported. The visit comes as US President Joe Biden’s administration seeks to ramp up Vietnam’s role in the global tech supply chain to reduce the US’ dependence on China. Images on

New apartments in Taiwan’s major cities are getting smaller, while old apartments are increasingly occupied by older people, many of whom live alone, government data showed. The phenomenon has to do with sharpening unaffordable property prices and an aging population, property brokers said. Apartments with one bedroom that are two years old or older have gained a noticeable presence in the nation’s six special municipalities as well as Hsinchu county and city in the past five years, Evertrust Rehouse Co (永慶房產集團) found, citing data from the government’s real-price transaction platform. In Taipei, apartments with one bedroom accounted for 19 percent of deals last

US CONSCULTANT: The US Department of Commerce’s Ursula Burns is a rarely seen US government consultant to be put forward to sit on the board, nominated as an independent director Taiwan Semiconductor Manufacturing Co (TSMC, 台積電), the world’s largest contract chipmaker, yesterday nominated 10 candidates for its new board of directors, including Ursula Burns from the US Department of Commerce. It is rare that TSMC has nominated a US government consultant to sit on its board. Burns was nominated as one of seven independent directors. She is vice chair of the department’s Advisory Council on Supply Chain Competitiveness. Burns is to stand for election at TSMC’s annual shareholders’ meeting on June 4 along with the rest of the candidates. TSMC chairman Mark Liu (劉德音) was not on the list after in December last