SciVision Biotech Inc (科妍生技), which makes hyaluronic acid for use in plastic surgery and the treatment of degenerative joint diseases, said yesterday that its revenue would grow more than 30 percent this year from last year, anticipating strong overseas sales of its degenerative joint disease treatments.
The company expects overseas sales to provide 64 percent of its revenue this year — up from 36 percent last year — as sales of its Hya-Joint degenerative joint disease treatment increased significantly in Malaysia, Turkey and Chile, SciVision Biotech general manager Tony Han (韓台賢) said yesterday.
The company also expects China to approve sales of its Hya-Dermis plastic surgery products this year, allowing the company to compete with Switzerland-based Q-Med AB, Korea-based LG Household & Health Care and one Chinese company, company chairman Han Kai-cheng (韓開程) said.
“The market for plastic surgery in China is a new and growing market with few competitors,” the chairman said.
SciVision is in talks with Chinese distributors and the company is likely to sign contracts with two firms to sell its products in the country, he said.
Regarding the Taiwanese market, the company expects sales to grow both monthly and annually starting next month after it launches a Hya-Dermis product with an anesthetic effect.
Gross margin for the new product is 15 percent higher than its other Hya-Dermis products, the company said.
Last year, the company reported revenue of NT$211.18 million (US$6.96 million), up 5.23 percent from NT$200.68 million a year ago, according to the company’s filing to the Taiwan Stock Exchange.
Hya-Joint products accounted for half of the company’s revenue last year, while Hya-Dermis products accounted for the other half, the company said, adding that it expects sales of Hya-Joint products to exceed 50 percent of its revenue this year.
Last month, SciVision registered revenue of NT$14.23 million, down 18.57 percent from NT$17.47 a year ago, according to filing.
Han Kai-cheng said revenue last month and this month would post year-on-year declines because customers are waiting for the company to launch its new products in Taiwan.
Shares in the company rose 0.85 percent to NT$71.1 yesterday, outperforming the TAIEX, which was down 0.48 percent.
Meanwhile, drugmaker TWi Pharmaceuticals Inc (安成藥) yesterday said it won a lawsuit in the US on Friday against US-based Par Pharmaceutical Inc, clearing patent infringement concerns for its generic drug Megace ES, which is used to treat eating disorders caused by chemotherapy or other treatments for HIV/AIDS.
The company said it would launch the drug by the end of the year and estimates about US$65.6 million a year in sales, which is 80 percent of what Par Pharmaceutical Inc gets for a drug treating patients with similar conditions.
TWi Pharmaceuticals’ share increased 2.2 percent to NT$325 yesterday.

Stephen Garrett, a 27-year-old graduate student, always thought he would study in China, but first the country’s restrictive COVID-19 policies made it nearly impossible and now he has other concerns. The cost is one deterrent, but Garrett is more worried about restrictions on academic freedom and the personal risk of being stranded in China. He is not alone. Only about 700 American students are studying at Chinese universities, down from a peak of nearly 25,000 a decade ago, while there are nearly 300,000 Chinese students at US schools. Some young Americans are discouraged from investing their time in China by what they see

Taiwan Semiconductor Manufacturing Co (TSMC, 台積電), the world’s largest contract chipmaker, yesterday reported record sales for the first quarter, which analysts attributed to solid demand for emerging technologies. Consolidated revenue totaled NT$592.64 billion (US$18.51 billion) in the January-to-March period, up 16.5 percent from a year earlier, but down 5.26 percent from the previous quarter, TSMC said in a statement. The first-quarter revenue beat analysts’ average projection of NT$579.5 billion, Bloomberg News reported. That performance lends weight to expectations that the world’s most valuable chipmaker would return to solid growth this year after weathering a post-COVID-19-pandemic cratering of smartphone and computer sales. TSMC is budgeting
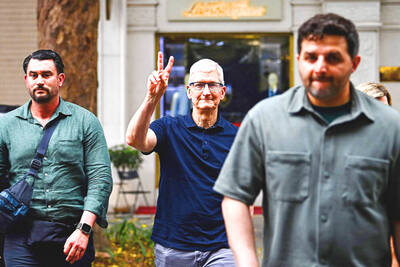
MAJOR DROP: CEO Tim Cook, who is visiting Hanoi, pledged the firm was committed to Vietnam after its smartphone shipments declined 9.6% annually in the first quarter Apple Inc yesterday said it would increase spending on suppliers in Vietnam, a key production hub, as CEO Tim Cook arrived in the country for a two-day visit. The iPhone maker announced the news in a statement on its Web site, but gave no details of how much it would spend or where the money would go. Cook is expected to meet programmers, content creators and students during his visit, online newspaper VnExpress reported. The visit comes as US President Joe Biden’s administration seeks to ramp up Vietnam’s role in the global tech supply chain to reduce the US’ dependence on China. Images on

US CONSCULTANT: The US Department of Commerce’s Ursula Burns is a rarely seen US government consultant to be put forward to sit on the board, nominated as an independent director Taiwan Semiconductor Manufacturing Co (TSMC, 台積電), the world’s largest contract chipmaker, yesterday nominated 10 candidates for its new board of directors, including Ursula Burns from the US Department of Commerce. It is rare that TSMC has nominated a US government consultant to sit on its board. Burns was nominated as one of seven independent directors. She is vice chair of the department’s Advisory Council on Supply Chain Competitiveness. Burns is to stand for election at TSMC’s annual shareholders’ meeting on June 4 along with the rest of the candidates. TSMC chairman Mark Liu (劉德音) was not on the list after in December last