ScinoPharm Taiwan Ltd (台灣神隆), which supplies pharmaceutical ingredients on a made-to-order basis, confirmed yesterday it would start offering ingredients to Vivus Inc this quarter for the sale of a new weight-loss pill in the US.
The Taiwanese firm’s confirmation came after the US-based Vivus announced overnight it had obtained approval from US regulators to sell the new drug, named Qsymia, and plans to start selling it by the end of the year.
“ScinoPharm announced today that its Tainan facility will provide commercial manufacturing of topiramate, the active pharmaceutical ingredient [API] for Vivus’ Qsymia, a new drug for the treatment of obesity approved by the United States Food and Drug Administration [FDA] on July 17, 2012,” the company said yesterday in a statement.
Qsymia is the second FDA-approved treatment for chronic weight management in adults in 13 years, after the US regulator on June 27 approved Arena Pharmaceutical’s anti-obesity pill Belviq.
For ScinoPharm, Qsymia is its fourth new drug approved by the US FDA, which it considers a more effective pill than Belviq.
Currently, ScinoPharm is working on more than 70 new projects with five entering Phase III clinical trials, according to the company.
Shares in ScinoPharm rose 1.23 percent to NT$57.7 in Taipei trading yesterday. The stock has risen 33.26 percent so far this year, compared with the TAIEX’s 1.08 percent increase over the same period. In US trading, Vivus surged 9.6 percent to US$29 on Wednesday.
ScinoPharm, located in the Tainan Science-based Industrial Park (台南科學園區), is the nation's largest API supplier to the global pharmaceutical and biotechnology companies. While serving the global generic industry, ScinoPharm also offers contract research and manufacturing services of existing and new drugs to companies around the world.
The company said its partnership with Vivus began with the supply of topiramate for the Qsymia clinical development program. ScinoPharm began commercial production of topiramate two years ago for the treatment of epilepsy and other nervous system disorders, it added.
With this approval for weight management, ScinoPharm president and chief executive Jo Shen (馬海怡) said in the statement that she expected the market potential for topiramate to increase substantially, without offering exact numbers.
Fubon Securities Investment Services Co (富邦投顧) analyst Heather Chang (張雅雯) said last month that Qsymia could become the world's most popular weight loss drug, after clinical studies showed that it can effectively assist in weight management without dangerous cardiovascular side effects.
"We forecast Qsymia can contribute between NT$100 million and NT$200 million to ScinoPharm's revenue this year and the sales of the drug will reach from NT$1 billion to NT$2 billion in five years," Chang said in a note on June 29.
ScinoPharm post NT$971 million in revenue in the first quarter of the year, down 13.9 percent from the fourth quarter of last year but up 22.2 percent from the same period of last year. Net income was NT$247 million in the first quarter, down 22.7 percent from the previous quarter but up 72 percent from a year earlier, with earnings per share (EPS) of NT$0.39.
Fubon forecast ScinoPharm's second-quarter revenue to show a sequential increase of 11 percent and an annual growth of 12 percent to NT$1.08 billion, while net income would grow 14 percent quarter-on-quarter and 10 percent year-on-year to NT$283 million, with NT$0.45 in EPS.
As the company's new production lines are due to come online later this year in Taiwan, coupled with contribution from the production of anti-depressant Viibryd and Galantamine for dementia, revenue is expected to rise 15.61 percent to NT$4.56 billion for the whole of this year and net income will grow 23 percent to NT$1.17 billion, with NT$1.86 in EPS, according to Fubon.

Stephen Garrett, a 27-year-old graduate student, always thought he would study in China, but first the country’s restrictive COVID-19 policies made it nearly impossible and now he has other concerns. The cost is one deterrent, but Garrett is more worried about restrictions on academic freedom and the personal risk of being stranded in China. He is not alone. Only about 700 American students are studying at Chinese universities, down from a peak of nearly 25,000 a decade ago, while there are nearly 300,000 Chinese students at US schools. Some young Americans are discouraged from investing their time in China by what they see
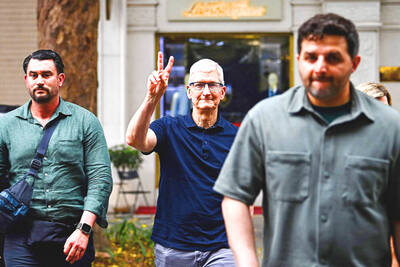
MAJOR DROP: CEO Tim Cook, who is visiting Hanoi, pledged the firm was committed to Vietnam after its smartphone shipments declined 9.6% annually in the first quarter Apple Inc yesterday said it would increase spending on suppliers in Vietnam, a key production hub, as CEO Tim Cook arrived in the country for a two-day visit. The iPhone maker announced the news in a statement on its Web site, but gave no details of how much it would spend or where the money would go. Cook is expected to meet programmers, content creators and students during his visit, online newspaper VnExpress reported. The visit comes as US President Joe Biden’s administration seeks to ramp up Vietnam’s role in the global tech supply chain to reduce the US’ dependence on China. Images on

New apartments in Taiwan’s major cities are getting smaller, while old apartments are increasingly occupied by older people, many of whom live alone, government data showed. The phenomenon has to do with sharpening unaffordable property prices and an aging population, property brokers said. Apartments with one bedroom that are two years old or older have gained a noticeable presence in the nation’s six special municipalities as well as Hsinchu county and city in the past five years, Evertrust Rehouse Co (永慶房產集團) found, citing data from the government’s real-price transaction platform. In Taipei, apartments with one bedroom accounted for 19 percent of deals last

US CONSCULTANT: The US Department of Commerce’s Ursula Burns is a rarely seen US government consultant to be put forward to sit on the board, nominated as an independent director Taiwan Semiconductor Manufacturing Co (TSMC, 台積電), the world’s largest contract chipmaker, yesterday nominated 10 candidates for its new board of directors, including Ursula Burns from the US Department of Commerce. It is rare that TSMC has nominated a US government consultant to sit on its board. Burns was nominated as one of seven independent directors. She is vice chair of the department’s Advisory Council on Supply Chain Competitiveness. Burns is to stand for election at TSMC’s annual shareholders’ meeting on June 4 along with the rest of the candidates. TSMC chairman Mark Liu (劉德音) was not on the list after in December last