Bristol-Myers Squibb Co, which failed to get US approval for a new diabetes treatment in January, will pay US$5.3 billion for Amylin Pharmaceuticals Inc, the maker of two drugs on the market for the disease.
The purchase comes a month after Bristol’s top seller, the blood-thinner Plavix with US$7.1 billion in sales last year, began facing generic competition. Next year, the New York-based company loses patent protection on its US$1.6 billion HIV drug, Sustiva.
Under the agreement announced on Saturday, Bristol-Myers will pay US$31 a share in cash, a 10 percent premium to Friday’s closing price for San Diego-based Amylin. At the same time, AstraZeneca PLC, based in London, will pay Bristol US$3.4 billion to help develop Amylin’s drug portfolio, the companies said.
It “looks a bit rich in terms of the price paid and it’s a trend in the sector, where biotech companies are commanding significant premiums, higher than they would have commanded in previous years because the pharmaceutical sector is being forced down this road,” said Navid Malik, an analyst with Cenkos Securities PLC in London.
The pharmaceutical industry lost patent protection on products valued at US$34 billion in annual sales last year, and revenue at risk from generics will rise to US$147 billion by 2015.
The diabetes market has become a key target for drugmakers as a result of rising obesity rates and the aging of the Baby Boom generation. About 346 million people globally have diabetes, and the number of deaths from the chronic disease may double from 2005 to 2030, according to the WHO.
AstraZeneca, Paris-based Sanofi and Merck & Co, of Whitehouse Station, New Jersey, also made offers during a bidding process, people with knowledge of the process had said.
Amylin ended a marketing deal with Indianapolis-based Eli Lilly & Co in November last year, and has been seeking a partner to sell Bydureon, a version of its diabetes drug Byetta, outside the US. The San Diego-based company began to seek acquisition suitors after rejecting a US$22-a-share offer from Bristol in February, people familiar with the matter said earlier this year.
For Bristol, the purchase is the largest of 19 since 2007, when it began a so-called string of pearls acquisition strategy designed to revitalize the company in the face of patent losses and produce a more diverse stable of products.
Bristol-Myers’s own experimental diabetes product, dapagliflozin, also called Forxiga, failed to win US marketing approval in January, when the Food and Drug Administration asked for more data to assess risks and benefits for the treatment, being developed with AstraZeneca. It is awaiting approval in Europe, and may be cleared later in the US.
The boards of Bristol-Myers and Amylin endorsed the deal, according to Saturday’s statement. Including Amylin’s debt and a payment owed to Eli Lilly & Co of about US$1.7 billion, the deal is valued at about US$7 billion.
“We are pleased to be able to strengthen the portfolio we have built to help patients with diabetes by building on the success Amylin has had with its GLP-1 franchise,” Bristol-Myers chief executive officer Lamberto Andreotti said.

Stephen Garrett, a 27-year-old graduate student, always thought he would study in China, but first the country’s restrictive COVID-19 policies made it nearly impossible and now he has other concerns. The cost is one deterrent, but Garrett is more worried about restrictions on academic freedom and the personal risk of being stranded in China. He is not alone. Only about 700 American students are studying at Chinese universities, down from a peak of nearly 25,000 a decade ago, while there are nearly 300,000 Chinese students at US schools. Some young Americans are discouraged from investing their time in China by what they see

Taiwan Transport and Storage Corp (TTS, 台灣通運倉儲) yesterday unveiled its first electric tractor unit — manufactured by Volvo Trucks — in a ceremony in Taipei, and said the unit would soon be used to transport cement produced by Taiwan Cement Corp (TCC, 台灣水泥). Both TTS and TCC belong to TCC International Holdings Ltd (台泥國際集團). With the electric tractor unit, the Taipei-based cement firm would become the first in Taiwan to use electric vehicles to transport construction materials. TTS chairman Koo Kung-yi (辜公怡), Volvo Trucks vice president of sales and marketing Johan Selven, TCC president Roman Cheng (程耀輝) and Taikoo Motors Group
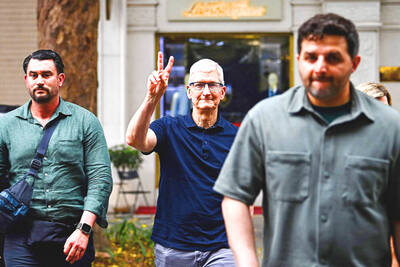
MAJOR DROP: CEO Tim Cook, who is visiting Hanoi, pledged the firm was committed to Vietnam after its smartphone shipments declined 9.6% annually in the first quarter Apple Inc yesterday said it would increase spending on suppliers in Vietnam, a key production hub, as CEO Tim Cook arrived in the country for a two-day visit. The iPhone maker announced the news in a statement on its Web site, but gave no details of how much it would spend or where the money would go. Cook is expected to meet programmers, content creators and students during his visit, online newspaper VnExpress reported. The visit comes as US President Joe Biden’s administration seeks to ramp up Vietnam’s role in the global tech supply chain to reduce the US’ dependence on China. Images on

New apartments in Taiwan’s major cities are getting smaller, while old apartments are increasingly occupied by older people, many of whom live alone, government data showed. The phenomenon has to do with sharpening unaffordable property prices and an aging population, property brokers said. Apartments with one bedroom that are two years old or older have gained a noticeable presence in the nation’s six special municipalities as well as Hsinchu county and city in the past five years, Evertrust Rehouse Co (永慶房產集團) found, citing data from the government’s real-price transaction platform. In Taipei, apartments with one bedroom accounted for 19 percent of deals last